[Image courtesy of Minnetronix]
The task of creating safe and reliable medical devices is the primary responsibility of medical device designers. Judgment must be used in assessing technology, diligence practiced in evaluating customer wants and needs, and completeness achieved in the execution of designs and processes. Integrating usability throughout the design lifecycle can greatly facilitate a favorable end result, leading to a safer device and better patient outcomes.
At Minnetronix, usability is fully integrated throughout the process, helping our customers avoid the pitfalls inherent in more piecemeal approaches.
The primary technical standard governing medical device design is IEC 60601-1, which defines general requirements for basic safety and essential performance. The 3rd edition of 60601-1 was published in 2005 and requires compliance with the IEC 60601-1-6 (which points to IEC 62366) usability standard. The FDA guidance “Applying Human Factors and Usability Engineering to Medical Devices” details how the FDA expects companies to meet these standards. Together, the guidance and the standards stress that good user design encompasses the entire design life cycle.
(Network with Minnetronix experts at DeviceTalks West, Dec. 11–12 in Orange County, Calif.)
Common pitfalls of neglecting usability
Numerous challenges can arise if usability is not considered throughout the process from early conception to final validation. These include:
- Early design but lack of follow-through: Often, companies undertake good early design work but neglect to ensure it is considered and/or implemented when it comes to writing code or performing mechanical design. For example, a team may hire an industrial design firm to interview end users and consider their needs at the onset of a project. However, as work progresses, such user findings can get swept away as personal opinions and old habits creep into the decision-making process.
- Late stage design changes: Conversely, companies may not consider users at the beginning of the project. They may find themselves having to attempt to adapt design in the later phases when alpha or beta prototypes are tested by users. Engineering teams often insist early on that they know what users want, only to be proven wrong by user feedback later in the process.
- Backfilling usability data: In order to meet technical standard requirements, companies may try to backfill usability efforts at the end of a project. Such belated usability is done only to pass the standards; there is no time or intent to make meaningful changes if issues are discovered. Sometimes, these efforts are undertaken post-release after problems that have resulted in adverse events.
- Beginning and end, but no middle: Occasionally, one firm is hired for initial design while a separate firm is engaged toward the end for usability testing, and a well-coordinated hand-off does not occur between the two. Testers are forced to re-accumulate background knowledge and potentially repeat work. Both designers and testers start from scratch, leading to wasted effort.
- Lack of integration: Another common scenario, particularly for smaller firms, is when usability is involved throughout, but it is being performed by a separate contract design firm. The outside vendor is not easily and fully integrated into internal requirements/FMECA/ verification. This disconnect often results in reduced efficiency and higher overall costs than full integration.
Optimizing usability integration for design life cycle success
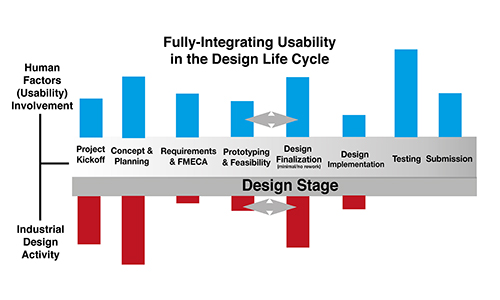
In order to avoid these and similar pitfalls, usability should be fully integrated throughout the process. Integration should include upfront contextual analysis; processes to understand client and end user needs; use of informed opinions to guide industrial, graphical, and user experience design; and usability personnel who are engaged throughout the design process. A clear understanding of human factors feeds into well-defined development tasks such as writing requirements and preparing the FMECA. Human factors need to be consistently incorporated during the project’s early, middle, and late stages.
Project initiation: Contextual analysis should include interviews with client stakeholders and field observation. Data should be collected on user groups, use environments, and tasks. This leads to a thorough understanding of end-user needs and constraints on the design, and it allows for creation of user needs based on real-world data. Such analysis informs design direction and helps come to a final concept. A team will be better able to create a task-based Use FMEA (UFMEA), which becomes the foundation of user testing.
Project development: The UFMEA and user needs drive design requirements. They provide evidence of reasoning for requirements and traceability from usability work into device design. The inclusion of a dedicated usability engineer on staff is critical; this staffer will help work through critical implementation details. The usability engineer will also serve as someone who always keeps the user in mind, asking questions such as “who is actually going to be using this, where will it be used, what do they really need? Having a dedicated usability resource will help mitigate feature creep and ensure improved safety of the design.
In terms of iterative formative testing, the involvement of a usability engineer means someone on the team tracks when it’s time for more user feedback. Well-timed testing allows the team to quickly make changes to the design before it is locked down. Rather than waiting on user testing until late in the process, thoughtful in-process testing allows the team to be more innovative with design.
Project conclusion: With usability personnel as part of the ongoing process, staff will be ready, onboard and informed for summative testing. Individuals who really know the project from beginning to end will be able to come up with more thorough and effective summative test protocols. They may be able to incorporate hooks to code and prototypes, allowing for more effective testing. In addition, early integration of user needs/UFMEA/task-informed requirements leads to a direct trace from the beginning to the end. This provides the firm with clear tracing from user needs to summative testing. Strong evidence will be in place that test requirements were clearly understood and appropriate testing was undertaken.
The Minnetronix approach
Minnetronix has developed a robust approach to the integration of usability. At each stage in the design life cycle, a focus on usability is a key component of the evolution of product design.
- User research: Here we define our user groups: how, where and when they will use the device, what tasks they perform, and what their core needs are. Information is gathered from stakeholder knowledge, research into predicate devices, identifying known use problems with similar devices, and field observations of users working with similar systems.
- Use specification: User research is compiled into a use specification. This includes a description of the users, use environment, and basic workflow. It is also where user needs are defined.
- Task analysis: This document includes a breakdown of user tasks and associated risks which feed into every element of the user interface design and evaluation process.
- UFMEA: User needs and task-associated risks from the use specification inform the writing of a use error FMECA.
- Design concepts and prototypes: Concepts for the device design are vetted against the knowledge summarized in the use specification. Prototypes of the system are generated to be evaluated by representative users.
- Formative testing: The task analysis is used to generate formative test procedures. Formative testing is performed with representative users on early prototypes or alpha versions of the devices. Formative testing is part of an iterative design process.
- Concept finalization and design specifications: As formative testing is completed, the design is finalized and knowledge gained is used to refine the user interface for the device. Gathered feedback, user needs, task analysis and the Use Error FMECA inform the writing of usability requirements and a user interface design document.
- Summative (usability validation) testing: When finalized devices become available, summative testing can be performed. A protocol is written based on tasks defined in the task analysis. The risk associated with various tasks in the use error FMECA determines pass/fail criteria. A submission quality summative report is generated which describes the entire usability process and the final evaluation.
Benefits of an integrated approach
Pursuit of such an integrated, user-centered design approach throughout all phases of the design life cycle has many valuable benefits, including:
- Improved product functionality and patient safety: Errors and field failures can be minimized due to careful testing of the design.
- Improved efficiency: Necessary changes are made during the life cycle at the appropriate time.
- Reduced costs: Numerous expenses can be avoided, including the costs of lost productivity, ill-timed design changes, implementation challenges, etc.
- Smoother regulatory pathways: Regulatory agencies can see the clear path from user needs to testing.
- Increased speed to market: Use of an integrative rather than a piecemeal process can expedite successful project completion and product commercialization.
- Higher user satisfaction and market acceptance: Customers are provided with well-designed, easy-to-use, safe products.
Sarah Blair is senior usability engineer at Minnetronix (St. Paul, Minn.). She has worked on more than 15 unique projects at Minnetronix, including those involving electrical stimulation devices, perfusion systems, patient-worn controllers for blood pumps and more.