Medtech developers can take advantage of middleware technology to gain a competitive advantage.
Tim Gee, Medical Connectivity Consulting
Two macro trends are affecting how medical devices and systems are designed: Adoption of off-the-shelf (OTS) computing technology to extend the power and capabilities of traditional embedded system medical devices; and the evolution of software architectures that greatly extend the ability to reuse software. Both of these trends have driven adoption of messaging middleware in the design of medical devices and systems.
As medical devices are transformed into networked information appliances, medtech manufacturers look to these two trends when designing central stations, remote surveillance, data analysis & diagnostic report generation and information gateway products. Rather than design these products as embedded system devices, industry best practice is to use OTS hardware and commercial IT software architectures. A common architecture is messaging middleware.
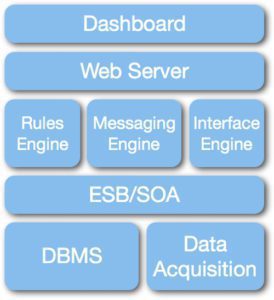
Messaging middleware is an amalgam of various software modules that use an enterprise service bus (ESB) to integrate and manage the modules. Key modules include a messaging engine, rules engine, interface engine, database, a web server and a data acquisition module to integrate the manufacturer’s medical device data. The selection of specific modules – there are many to chose from – and the inclusion of any additional modules depends on the project’s market requirements.
A common example of this architecture is the Mirth Connect interface engine, which is designed around its own ESB and includes messaging, rules, database and web server modules. A competitive patient monitoring central station has been developed using messaging middleware architecture. This product was developed in less than 12 months with 7 engineers and received FDA clearance in less than 100 days using mostly open-source software.
Both commercial and open-source modules are available for building this software architecture. Vendor support is available for most of the open source modules.
This approach impacts manufacturers in a number of important ways. By leveraging messaging middleware architecture, manufacturers can come to market in a much shorter period of time compared to conventional software development methodologies. This rapid time-to-market translates to substantially lower overall product development costs.
By integrating feature-rich modules into an overall architecture, it’s possible to launch an initial product with a substantially greater set of features. Once the modules are integrated, virtually all product functionality comes from how the various modules are configured, rather than adding to or changing the source code of the underlying architecture. Consequently, product changes and enhancements require less time and skill.
The trade-off for these benefits centers on changes required by the manufacturer to effectively support the development of a product based on a messaging middleware architecture. These changes hit the engineering, quality and regulatory departments. The degree of these changes depends on how far along a company is on the journey to adopt commercial software development methodologies for their non-embedded system products.
The development tools and engineer skillsets differ substantially between embedded system software development and development using the commercial computing platform software approach required by messaging middleware. This change can require investment in tools, engineer training, and perhaps, bringing in engineering resources with new skills.
The quality department will see new verification testing requirements for messaging middleware compared to embedded systems devices. Due to the substantial functionality available in these types of architectures, automated software testing, commonly used for commercial software, will have a big impact on minimizing the common testing bottleneck for manufacturers’ product development process.
The final opportunity for change driven by messaging middleware lies in regulatory affairs. There are several potential levers where messaging middleware based products can foster competitive regulatory strategies, especially impacting sustaining engineering (development after initial product introduction). The way requirements are written and tested can minimize the scope of verification testing. The need to submit new 510(k)s can also be reduced as a consequence of messaging middleware based product features being implemented in software configuration rather than writing source code.
Messaging middleware software architectures are increasingly adopted by medtech due to significant competitive advantages over older architectures. To realize the full benefits requires manufacturers to make adjustments to their operations and, perhaps, human resources. The areas most impacted are engineering, quality and regulatory affairs. Because messaging middleware is a mature technology, this transition is more about navigating successful organizational change across multiple departments than it is about the technology itself.
Tim Gee is principal and founder ofMedical Connectivity Consulting (Beaverton, Ore.), focused on workflow automation through the integration of medical devices with information systems and enabling technologies.