It’s tough to pick just 10 medtech startups to profile out of the hundreds or thousands dotting the U.S. landscape, but we tried anyway.
It helped that we kept our criteria simple: A relatively new company, with only one or two (or no) financing rounds under their belts. Some are so deeply in stealth mode that they never responded to our enquiries.
More important in our selection process was that the technology being developed sparked our interest. That made it even tougher to winnow the field, as the array of unmet needs being met by ingenious medical devices is truly inspiring.
In the following pages you’ll find brief looks at our choices, ranging from the radiofrequency ablation device for treating overactive bladder being developed by stealthy Amphora Medical, to Sonex Health’s Stealth Microknife device for treating carpal tunnel syndrome.
And to all those startups who didn’t make the cut: It’s not you, it’s us – we only had 10 slots and there are so many of you! Don’t worry, there’s always next year…
![]() Amphora Medical
Amphora Medical
Stealthy Amphora Medical is stingy with details about the technology it’s developing to treat overactive bladder. Founded by serial entrepreneur Danny Sachs and medtech veteran Ed Hlavka, Amphora’s single-page website offers only contact information and a link to a lone patent.
 But the company has managed to raise nearly $13 million for the device it’s developing to treat overactive bladder. According to the patent cited on its website, Amphora’s device is designed to ablate nerves in the bladder’s trigone, the funnel-shaped portion that leads to the urethra. The tech aims to denervate a portion of the trigone without disturbing the bladder’s outer mucosal layer, according to the patent.
But the company has managed to raise nearly $13 million for the device it’s developing to treat overactive bladder. According to the patent cited on its website, Amphora’s device is designed to ablate nerves in the bladder’s trigone, the funnel-shaped portion that leads to the urethra. The tech aims to denervate a portion of the trigone without disturbing the bladder’s outer mucosal layer, according to the patent.
Amphora is running a pair of trials evaluating the device, according to ClinicalTrials.gov. The Caret-I trial, a Phase I/II study of 50 patients with a primary endpoint of device-related complications at 12 weeks, is slated for final data collection in October 2016. The Caret-II trial, a 2-phase safety and efficacy study, is designed to evaluate device-related serious complications at 4 weeks. It’s final data collection is due in June 2016.
Amphora came up with $600,000 in a hoped-for $1.25 million equity round involving 4 investors back in 2013, according to SEC filings. The round was expanded to nearly $12.8 million last year, the filings show.
Amphora did not respond to our attempts to contact them, but we’ll certainly be keeping an eye on this intriguing startup.
Maple Grove, Minn.
AmphoraMedical.com
![]() Aum Cardiovascular
Aum Cardiovascular
• Aum Cardiovascular is developing the CADence handheald heart disease diagnostics device, designed to identify obstructions in the coronary arteries by detecting the acoustic signals generated by turbulence created as blood flows past an obstruction.
• Founded in 2009 by Marie Johnson
• SEC-registered funding – $456,000, $3.3 million in 2011; $1.8 million in 2013; $5,006,150 in 2015 – TOTAL: $10,562,150
Marie Johnson was working on a prototype computerized stethoscope device designed to listen to heart sounds as part of her doctoral degree when her seemingly healthy, 41-year-old husband died suddenly from a heart attack.
Johnson’s husband had recently been given a clean bill of health after undergoing a traditional stress test, which made the cause of his death even more shocking. She later found out that he had blockages in his coronary arteries, including ruptured plaque in the left anterior descending artery – the aptly named “widowmaker.” Her husband’s own coronary artery disease had gone undetected by traditional tests.
“I had 2 young children,” Johnson says. “I knew right then I was going to work on eradicating this terrible disease.”
So she set to work putting the principles of frequency analysis she’d used in her doctoral program to create an acoustic device to identify obstructive coronary artery disease.
The device, later named CADence, would become the cornerstone of the company Johnson founded, Aum Cardiovascular.
The name is based on a Sanskrit syllable meaning “to make a continuous low humming sound,” akin to the information the CADence device is designed to extract from diseased coronary arteries, the company says.
Johnson got the patent for her computerized stethoscope back from the University of Minnesota, where she’d developed it, and set out to start Aum Cardiovascular with a relatively small grant from the federal government.
A mentor – medtech legend and serial entrepreneur Dr. Manny Villafaña – suggested she seek out private placements to get the company running. That’s helped Aum bring in about $10.3 million. It wasn’t just the story, but also the effectiveness of the device, that wooed investors, Johnson says.
“They don’t give you money because they’re in love with the story,” she explains. “They funded this because they saw that we could detect obstructive coronary disease with an easy test, and with no pharmaceuticals. It was a no-brainer.”
Normal blood flow through coronary arteries is smooth and even; arterial blockages disrupt that smooth flow and create turbulence. That turbulence creates a particular noise pattern that’s far too faint to be heard with a traditional stethoscope.
Using sound to identify coronary artery disease isn’t a new idea – in 1967, an article in the American Journal of Medicine by William Dock and Samuel Zoneraich detailed a “high-frequency diastolic murmur” found along the left parasternal area at the 2nd, 3rd or 4th intercostal space. The sound was correlated with arterial stenosis, and would go on to be referred to as “Dock’s Murmur.”
In a similar manner, the CADence system “listens” for the sounds of disrupted blood flow in diseased coronary arteries. A test with the device takes approximately 20 minutes to perform, and requires minimal training to conduct. The CADence system collects data from four thorax wall locations; afterward the collected data is uploaded via Bluetooth to Aum Cardiovascular’s proprietary analytics engine.
A report based on the uploaded is later delivered via email to either the clinician or another account determined by the clinician.
The test is cheap compared to standard nuclear stress tests, which come with a $5,000 tab. Each test with the CADence device costs only $100.
The anecdotal also helps Aum, as one investor actually credits the device with saving his life. At one of Johnson’s investor pitch sessions, the Aum device was used to scan Linn Grove Ventures CFO Steve Kiemele and indicated that he might have a 60% to 70% blockage of the left anterior descending artery.
The blockage was later confirmed by a cardiologist, and although it was not as extensive as indicated by the CADence device, the revelation was enough to “shock” 51-year old Kiemele, who Johnson describes as athletic and not overweight.
Last November, Aum finished enrollment in 1,000-patient non-inferiority study of the device, comparing it against nuclear stress tests in subjects who present with chest pain and two or more cardiac risk factors, and are indicated for nuclear stress testing to determine if they have coronary artery disease.
Aum won CE Mark approval in the European Union in November 2014 and launched in Germany in 2015.
Northfield, Minn.
aumcardio.com
 Bigfoot Biomedical
Bigfoot Biomedical
• Bigfoot Biomedical is developing an artificial pancreas, using a combination of modern technologies such as continuous glucose monitors, insulin pumps and proprietary algorithms.
• Founded in 2014 by former Junior Diabetes Research Foundation CEO Jeffrey Brewer, home-CGM hacker Bryan Mazlish and ex-Medtronic chief engineer Lane Desborough.
• SEC-registered funding: $3 million, $3.75 million, $6.5 million, 2015; $2,375,000, 2016. TOTAL: $15,625,000
In 2014 Bryan Mazlish made waves as the stealthy subject of a Wired article that tagged him as the first developer of a home-brewed bionic pancreas.
The article referred to Mazlish as “Bigfoot,” as Mazlish was staying under the radar with his hacked device, designed to automatically manage both glucose testing and insulin delivery to manage the Type I diabetes afflicting both his child and his wife.
Mazlish, at the time a Manhattan stock-trading programmer, had a wealth of experience working with computer algorithms designed to autonomously buy and sell stocks. He used his expertise to develop an algorithm to manage Type I diabetes, in a quest to give them more freedom from the chronic condition.
The “Bigfoot” name stuck, later to be used as the moniker for the company Mazlish helped found: Bigfoot Biomedical.
The initial system operated on a hacked-together combination of an insulin pump, a continuous glucose monitor and a smartphone, using proprietary algorithms to determine insulin dosage based on glucose readings. Mazlish’s wife later reported that she and their son had been using the closed-loop system for more than 2 years with no issues, touting an impressive 30,000 hours on the system.
Mazlish teamed up with ex-JDRF chief Brewer and ex-Medtronic engineer Lane Desborough to found Bigfoot. The company’s goal is to bring a fully functional, automated insulin delivery system to the market with a sustainable business model. The company, which has kept a low profile since the Wired article, says it’s already assembled a prototype automated insulin delivery system that uses the patient’s basal insulin dose hypoglycemia susceptibility to gauge the timing and size of dosages.
The system is also designed to allow patients to tell it how many carbohydrates they’re eating, to allow it to calculate a bolus dose. Otherwise it’s designed to manage itself, leaving patients free from worry until their next meal or site change.
In May 2015, Bigfoot picked up the assets of shuttered insulin pump maker Asante Solutions and its FDA-cleared Snap insulin pump, planning to integrate the pump into its smart delivery system. The buyout also meant a big move for Bigfoot, which swooped in to take over Asante’s headquarters in Milpitas, Calif.
Only a month later, Bigfoot was on the prowl again, inking a deal with continuous glucose monitor industry giant Dexcom to integrate data from its CGM systems into the Bigfoot smart delivery system, as with Mazlish’s original prototype.
Bigfoot Biomedical hopes to have a clinical study under way late this year. The so-called “hotel study” is designed to allow patients to use the artificial pancreas in a real-world setting, but with staff on hand at all times.
A large, 3-month study of the device is planned for early 2017, looking to enroll a few hundred patients as the company continues its quest for regulatory clearance.
Bigfoot says it’s aiming for an FDA submission by the end of 2016, with a hopeful date for clearance and market entry by the end of 2018.
The company has already raked in over $15 million in funding, starting in June last year, and has set its sights on acquiring another $15 million.
Milpitas, Calif.
bigfootbiomedical.com
Cardialen
• Raised a total of $6.8 million, according to regulatory filings, plus grants and loans
• Device is based on low-energy cardioversion technology licensed from Washington University in St. Louis and prior Case Western Reserve University research.

Chronically Implanted Lead Positions and Experimental Timeline Fluoroscopic images of the anatomic positions of chronically-implanted transvenous leads and subcutaneous (SC) access ports from left lateral (left panel) and left anterior oblique (right panel) views.
Cardialen is developing a low-energy implant for atrial fibrillation designed to control the fibrillation without damaging heart tissue. The technology uses series of lower-energy pulses rather than a single high-energy shock to effect the therapy.
Pre-clinical testing shows that the Cardialen device significantly reduces the atrial defibrillation threshold compared with a single shock. A clinical feasibility study is under way and slated to be complete this year.

Schematic depiction of lead positions. Shocks were delivered from the right atrium (RA) coil to the left pulmonary artery (LPA) coil or the RA coil to the coronary sinus (CS) coil.
The company said it’s designing the system using existing implantable device “building blocks,” including commercially available cardiac leads, so that “the Cardialen therapy system can be rapidly adopted using existing care pathways, procedures, reimbursement, and managed using existing infrastructure.”

Experimental timeline showing model development and approximate times of defibrillation (defib) studies.
Cardialen is led by chairwoman & CEO Paula Skjefte, a veteran executive from Medtronic, and vice president of development Brent Shelton, also a Medtronic alum.
Minneapolis & St. Louis
cardialen.com
InterValve Inc.
• Founded 2008 by Mark Ungs and William Drasler
• Raised $4.7M A round in 2011; Debt round of $3M in 2015
InterValve is developing a next-generation aortic valvuloplasty balloon for treatment of calcific aortic stenosis. Founded in 2008 by an experienced group of cardiologists and industry executives, Intervalve is developing tools for the percutaneous and transapical aortic valve replacement procedures.
CEO Mark Ungs served as the vice president of new business development for Boston Scientific’s interventional cardiology division during the development of drug-coated stents and cardiac resynchronization therapy for CHF, the two biggest blockbuster devices in the history of the medical device industry. Ungs is also the co-founder of ACTx, a startup developing a therapy for tissue function recovery and regeneration.
 InterValve’s key technology is the V8 aortic valvuloplasty balloon catheter, a figure-8-shaped device designed to treat calcification of the aortic valve. For the core patient, often elderly and at high risk for mortality from traditional open heart surgery, surgical valve replacement surgery is highly invasive and usually requires a long recovery period. An estimated 50% of patients with aortic stenosis are therefore not eligible for or choose not to have this valve replacement surgery.
InterValve’s key technology is the V8 aortic valvuloplasty balloon catheter, a figure-8-shaped device designed to treat calcification of the aortic valve. For the core patient, often elderly and at high risk for mortality from traditional open heart surgery, surgical valve replacement surgery is highly invasive and usually requires a long recovery period. An estimated 50% of patients with aortic stenosis are therefore not eligible for or choose not to have this valve replacement surgery.
The shape of the V8 device is important because it’s designed to lock into the atrial valve’s anatomy. In conventional catheter balloon procedures, it’s difficult to determine the precise amount of dilatation force exerted on the aortic valve annulus that’s needed to restore its conformity. Given this limitation, the tendency is to undersize the balloon in an attempt to limit complications. But in doing so, the operator potentially accepts some procedural compromises, such as sub-optimal valve area gain and a higher rate of balloon slippage.
Limiting movement of the balloon reduces procedure and ischemic time. The InerValve device solves the problem of balloon slippage when dilating the aortic valve during valvuloplasty, either as its own procedure or in preparation for transcatheter aortic valve replacement (TAVR). The balloon’s center is positioned at the valve’s opening with the help of a radiopaque marker. Once aligned, the balloon is inflated, taking on its namesake figure-8 shape.
The company won FDA clearance in 2013 for the V8 device. In October 2014, the catheter was successfully used to dilate the aortic valve annulus during a TAVR procedure guided by live 3D transesophageal echocardiography. In January 2015, the company closed a $3 million senior secured term loan agreement with Oxford Finance LLC to continue commercial and development efforts. It also named Maquet Medical Systems USA as its sole US distributor.
Plymouth, Minn.
intervalveinc.com
- Founded in 2001 by Srirama Rao; Jim Buck is CEO
- Raised $9.38 million in 2013.
Functional mitral valve regurgitation (FMR) is a bit of a misnomer, because it’s considered a disease of the left ventricle, not of the mitral valve. FMR occurs when the heart’s left ventricle is distorted or dilated, displacing the papillary muscles that support the valve’s two leaflets and stretching its annulus and allowing blood to flow back into the atrium. If left untreated, FMR overloads the heart and can lead to or accelerate heart failure.
For more than 30 years the standard of care has been a valve repair procedure called annuloplasty, which involves the implantation of a ring or band around the annulus to restore its natural shape. But that surgical technique, and the annuloplasty ring, have gone nearly unchanged over the last decade.
What’s more, annuloplasty is challenging to perform and very invasive (open-heart and on bypass), and because the cause of FMR is not addressed, regurgitation can reoccur in 40% or more of patients. Because the surgical risks are significant and there is a high rate of reoccurrence, only a small fraction of indicated patients undergo surgical procedures for FMR every year.
Mardil Medical’s VenTouch system targets the root cause of FMR by applying light pressure to push the valve leaflets closer together, stopping the regurgitation. It’s also designed to provide gentle support to the ventricular wall to prevent further distortion, which can lead to the reccurrence of FMR.
The VenTouch device is a sleeve that slips around the heart (pictured) and inflates with saline until the mitral valve annulus has been coaxed into conformity. Should further changes in the left ventricle occur after implantation, the system can be adjusted using a remote port.
First-in-human implants came in February 2014 at the Institut Jantung Negara National Heart Institute in Kuala Lumpur in two patients as part of a 15-patient Phase I study. The primary safety endpoint is the rate of serious adverse events at 6 months, with final data collection slated for June 2017.
mardil.com
MIVI Neuroscience
MIVI Neuroscience is developing a catheter-based treatment for acute ischemic stroke that’s designed to extract the blood clots that cause it from the brain.

Illustration of an ischemic stroke, which occurs when a brain blood vessel gets blocked. The gray area represents brain tissue that is not receiving nutrients as a result of the stroke.
Founded in May 2013 by president Matt Ogle, the company won 510(k) clearance from the FDA for a 6Fr version of its Mi-Axus catheter a scant 18 months later. MIVI Neuroscience says it expects to launch the rest of the system’s components over the next two years and anticipates winning CE Mark approval in the European Union in the next year for the Neuronet embolic protection component.
The Mi-Axus catheter tapers from 6Fr to 5Fr and features a flexible shaft that’s designed to minimize arterial trauma during clot retrieval. A braided catheter body offers improved torque and navigation and has a hydrophilic coating designed to reduce friction as the catheter travels through blood vessels.
MIVI Neuroscience reeled in $2 million in August 2015 in a seed financing round, plus $185,000 in debt financing from Minnesota’s Angel Loan Fund, saying it planned to use the cash to optimize its technology and fund initial clinical work.
mivineuro.com
- Raised a $5 million Series B round in March 2015
- Won FDA clearance in 2013, after just 4 years, because its device could be tested in the lab rather than in a clinical trial
Pursuit Vascular developed its ClearGuard technology to address a particularly pernicious unmet need: Protecting patients from bloodstream infections stemming from long-term catheter and port use.
 It’s a deceivingly prosaic indication, but catheter-related bloodstream infections are one of the most common – and lethal – complications from central venous catheterizations. It’s estimated that as many as 10% of central venous catheterizations lead to CRBSIs, which have a mortality rate of 25% and significantly boost both length of stay and treatment cost. The infections cause about 30,000 deaths in the U.S. each year, adding an estimated $6 billion to healthcare costs.
It’s a deceivingly prosaic indication, but catheter-related bloodstream infections are one of the most common – and lethal – complications from central venous catheterizations. It’s estimated that as many as 10% of central venous catheterizations lead to CRBSIs, which have a mortality rate of 25% and significantly boost both length of stay and treatment cost. The infections cause about 30,000 deaths in the U.S. each year, adding an estimated $6 billion to healthcare costs.
The FDA-cleared ClearGuide HD device is aimed at the hemodialysis market. It’s an anti-microbial end cap for which the federal safety watchdog created a new product code covering its “use in hemodialysis catheters to reduce hub infection.”
The cap works via a rod coated with the anti-microbial chlorhexidine that extends into the hemodialysis catheter hub. The coating is activated when the end cap is inserted into a liquid-fileld catheter. The dissolved chlorhexidine is held inside the catheter by the existing catheter clamp; the amount of the agent released is controlled to ensure safety even if the clamp is opened.
Pursuit Vascular cites independent testing showing that ClearGuard HD reduces an array of nasty bugs – including varieties of staphylococcus, enterococcus, e. coli and candida – by more than 99.99 percent.
Largely financed by angel investors in the Twin Cities, Pursuit raised a $5.1 million Series B round in March 2015. The company was co-founded by R&D vice president Bob Ziebol in 2009 and is led by president & CEO Doug Killion.
Proceeds from the Series B round are slated, in part, for scaling up production of the ClearGuard HD device.
Maple Grove, Minn.
pursuitvascular.com
Sonex Health
Carpal tunnel syndrome affects more than 12 million Americans and results in 500,000 surgeries each year. The traditional fix has been an invasive surgery that involves cutting the transverse carpal ligament in the wrist, which is not expansible. The ligament forms the roof of the carpal tunnel and can pinch a nerve in the wrist causing the discomfort experienced in carpal tunnel syndrome patients. The conventional solutions expand the space around the nerve.
“That is done by expanding the tunnel – cutting the ligament over the nerve,” says CEO Dr. Darryl Barnes. “In the procedure, you cut the skin, dissect down to the ligament, transect the ligament and then sew up everything you cut, except for the ligament. It is considerably invasive and not as safe as it should be.” And while this remedies the condition, it comes with drawbacks, such as large and sometimes painful scars, ongoing palmar pain, and a long road to recovery.
The ultra-low-profile Stealth MicroKnife device uses technology developed at the Mayo Clinic to address carpal tunnel syndrome.
“Our inspiration was to make it less invasive, less expensive, and safer to improve the lives of those who suffer from it. Dr. Jay Smith and I had developed several products around ultrasonically guided procedures and came up with the idea for the MicroKnife a few years ago,” says Barnes.
The first MicroKnife prototypes were able to reduce the size of the traditional incision from two in. to about ½ in. (12.7mm). A few prototypes later, the device now requires an incision of 3mm to 5mm. “The MicroKnife is much smaller than the illustration on our website makes it appear. For example, it is only 2.8mm high.”
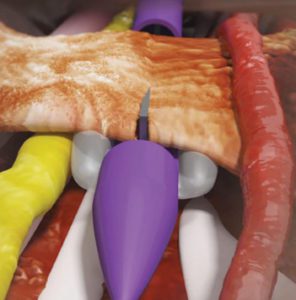
The Stealth MicroKnife (the thin grey wedge) is position while sheathed. Other device features protect surrounding nerves and vessels.
Once properly positioned using ultrasound, the device provides a “safe zone” barrier to protect nerves, blood vessels, tendons, and other sensitive anatomic structures during transection. The instrument will let physicians perform carpal-tunnel-release surgery through a single micro-incision with ultrasound guidance (or with a traditional mini-open incision without ultrasound guidance) while protecting sensitive anatomy during transection of the transverse carpal ligament. In fact, the Sonex says, MicroKnife requires an incision so small it can be closed with a small adhesive bandage or strip instead of sutures.
Immobilization is unnecessary, so patients can begin rehabilitation and get back to their jobs and lives almost immediately. The result is a low-trauma outpatient procedure associated with a recovery that is shorter than that of conventional surgical methods.
What’s more, the procedure formerly took place in an operating room. The innovation will let physicians safely and effectively perform carpal tunnel release in an office or ambulatory surgery center, resulting in the rapid patient recovery, improved cosmesis, and reduced costs.
The company was founded in 2014 by Barnes and Smith, both physicians at Mayo Clinic, and business operations expert Aaron Keenan. Regarding funding, Barnes and Keenan, who serves as CFO, would only say the private company raised sufficient capital by July 2015 and is funded to reach key milestones, such as inpatient use.
The Stealth MicroKnife is not for sale yet. Prototypes have been successfully tested on cadavers multiple times and gone through several iterations of testing and redesign. “We recently froze the design and are now in the manufacturing process of creating the device,” says Barnes.
To find its way along the regulatory path, Keenen says the company has taken guidance from a reputable consulting firm. Sonex hopes the MicroKnife will be registered with the FDA as a Class I device under a 510(k) clearance.
“To meet the regulatory requirements, we have done rigorous testing with cadaver studies and will be performing final instrument test including biocompatibility, sterilization, and more. Presently, we are setting up manufacturing and will have to meet all regulations for good manufacturing principles, and prove that we designed our instrument with good manufacturing principles. The Stealth MicroKnife has been designed and is fully complaint with ISO 13485 and 14971 standards,” says Keenan.
The first human patient is scheduled to undergo the procedure during the fall of this year.
Rochester, Minn.
sonexhealth.com
TriFusion Devices
• Founded in 2014 by Texas A&M grad student Blake Teipel and Brandon Sweeney
• Won nearly $400,000 in business competitions
 Some of the most exciting start-ups these days are coming out of universities. Take TriFusion Devices, an up-and-coming company that just won the coveted Rice Business Plan Competition. The nascent company’s technology has won nearly $400,000 in business competitions for the proprietary microwelding process behind its 3D-printed prosthetic devices, illustrating its appeal as easy to understand, filling an unmet need and offering time and cost savings.
Some of the most exciting start-ups these days are coming out of universities. Take TriFusion Devices, an up-and-coming company that just won the coveted Rice Business Plan Competition. The nascent company’s technology has won nearly $400,000 in business competitions for the proprietary microwelding process behind its 3D-printed prosthetic devices, illustrating its appeal as easy to understand, filling an unmet need and offering time and cost savings.
TriFusion’s leadership comes out of Texas A&M’s Materials Science & Engineering department; CEO Blake Teipel is a Ph.D. candidate with a background in product and business development; CTO Brandon Sweeney is also a Ph.D. candidate with experience in nanotechnology development and 3D printing for robotic vehicles; and Britton Eastburn, a medical student at Texas A&M’s HSC College of Medicine, serves as the chief medical business officer.
Creating a prosthetic can cost almost $30,000 and take at least a month, for a device that will last only 5 years. TriFusion developed a carbon nanotube-coated printer filament and a patent-pending microwave welding process to fuse 30-pin parts. The unique property of the carbon nanotubes overcomes the difficulty of the weld strength of polymers, enabling stronger 3-D printed prosthetics at a fraction of the cost of currently available devices.
“The microwelding process is a disruptive technology, due to an enormous competitive advantage in both price and production time,” Sweeney explains.
Using this technology, the TriFusion team says it can slash a long and expensive process by going from scan to device in less than 48 hours. Eastburn says they can produce 3D-printed parts with mechanical properties that rival conventional prosthetics and also enable post-printing fit adjustment for the final socket, eliminating the traditional limb-casting process and the need for test-fit sockets.
And the team sees larger applications in the medical arena. “We can make prosthetics, orthotics, wearables, sporting equipment and protective devices,” says Eastburn.
But it’s early days yet. TriFusion is looking to raise $1.8 million to take its prosthetics technology to market.
College Station, Texas
trifusion.co




![A photo of the Medtronic GI Genius ColonPro polyp detection system flagging a potential sign of colon cancer during a colonoscopy. [Photo courtesy of Medtronic]](https://www.medicaldesignandoutsourcing.com/wp-content/uploads/2024/04/Medtronic-GI-Genius-doctors-268x170.jpg)


