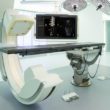
Philips’ augmented reality surgical navigation technology is designed to help surgeons perform image-guided open and minimally-invasive spine surgery. [Image from Philips]
The company is touting its advanced interventional imaging systems, diagnostic and therapeutic devices, planning and navigation software and various services. It is also showcasing its latest cardiac care solutions for ultrasound and image-guided therapy. Its Dynamic coronary Roadmap 2.0 with FFR/IFR Roadmap is also on display at the event.
Medtech giant Royal Philips has been working on image-guided therapies for the last year, hoping to create cost-effective healthcare. The company released its Azurion imaging platform for low-dosage X-rays along with its augmented reality (AR) surgical navigation technology earlier this year. Philips suggests that image-guided therapies can help bring a more personalized treatment system to patients.
A combination of imaging systems, interventional devices, navigation tools and patient health records helps Philips’s Azurion give physicians the control and information they need to curb cardiovascular disease, according to the company.
Interventional X-ray systems from Philips are joined by the company’s new and next-generation 3D TEE technology that allows physicians to have a new level of live 3D imaging. It has a new acoustic design that features higher frequencies and bandwidth to allow for better resolution and tissue fitting.
The company has also launched its diagnostic device, the Refinity ST next-generation rotational intravascular ultrasound (IVUS) catheter. It features exceptional deliverability, 5F guide compatibility and 45 MHz high-resolution imaging. Refinity ST helps clinicians with complex percutaneous coronary interventional cases.
Philips’s CoreVision iFR co-registration solution and the CoreIVUS and physiology technology helps physicians in the stunting process when lesions are more complex.
Philips is also touting its Stellarex drug-coated balloon therapeutic device at TCT this year. It is the only commercially-available drug-coated balloon and has been shown to have durability and consistently high potency rates in a variety of patients.
“TCT provides the opportunity for us to share our expanded portfolio that aims to help those affected by cardiovascular disease to get back to living their lives,” Bert van Meurs, business leader of Image Guided Therapy for Philips, said in a press release. “At the congress this year, we’ll demonstrate how we’re furthering our goal to help clinicians decide, guide, treat and confirm the right therapies for their patients and provide safe and personalized treatments to enable faster recoveries while reducing costs.”
The company is presenting its technology at booths 1029 and 4133 at TCT in Denver, Oct. 29 through Nov. 2.