Metal-on-metal hips

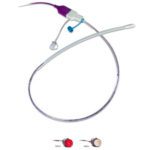
Depuy ASR XL hip replacement [Image from Depuy]
As of the end of last year, J&J still faced lawsuits from 10,500 plaintiffs with direct claims related to the Pinnacle acetabular cup system, as well as 1,800 directly affected by the DePuy ASR XL acetabular system and DePuy ASR hip resurfacing system, according to J&J’s most recent annual report.
Other device makers have found themselves pulled into the controversy since 2010. Even metal-on-metal implants that haven’t undergone a recall have become embroiled in personal injury lawsuits.
In 2013, the U.K.’s National Institute for Health & Care Excellence found failure rates as high as 43% for some metal-on-metal implants. Worse, reports have warned of hundreds of thousands of patients potentially exposed to toxic compounds from the implants, putting them at risk of developing cancer, cardiomyopathy, muscle and bone destruction and changes to their DNA. One study found that metal ions that were released from the cobalt-chromium-molybdenum metal devices when they rubbed together would penetrate bone marrow and impede bone-growing cells from forming.