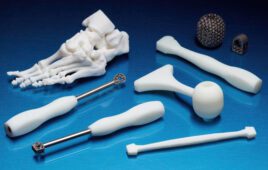
Medical prints made with Formlabs 3D printers [Photo courtesy of Formlabs]
3D printer accessibility and affordability are enabling small device firms to develop and market a wave of personalized devices.
Gaurav Manchanda, Formlabs
Healthcare is becoming more efficient, and patients are beginning to expect a personalized approach. 3D printing isn’t a newly minted manufacturing technology, yet it’s reached an inflection point for bringing change in healthcare and dental applications.
Traditionally, 3D printing was prohibitively expensive and only available to the largest, best-resourced medical centers and device manufacturers. But these days, 3D printers have become more affordable and accessible. As a result, additive manufacturing is surging in healthcare as medical providers and device manufacturers tap into the ability to safely produce novel, patient-specific, biocompatible and sterilizable parts.
As 3D printing becomes the method of choice for a growing number of medical devices, implants, surgical guides, orthotics, prosthetics, and other medical applications, medical device firms and manufacturers must consider the technology for the commercialization of end-use parts.
Related: What’s new in 3D printing: medical devices, research, innovation, automation and partnerships
The road to 3D printing commercialization in healthcare

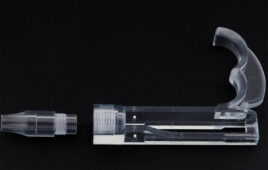
Medical products made with SLS 3D printing [Photo courtesy of Formlabs]
3D printing manufacturers and healthcare leaders are teaming up to further advance additive manufacturing. Major group purchasing organization (GPO) Vizient listed a 3D printing company in its catalog for the first time, and other partnerships are accelerating the delivery of life-changing medical devices while maintaining regulatory compliance.
This support from the industry dovetails with advancements enabling new applications and faster workflows, which will further advance 3D printing toward commercialization in healthcare. Beyond 3D printing hardware and software, materials are also a critical component to advancing 3D printing commercialization in the space.
3D-printed medical products made with BioMed resins [Photo courtesy of Formlabs]
Designing 3D printing technology with healthcare commercialization in mind will enable the industry to reach the end goal of improving patient care sooner. Small and large medical manufacturers have started creating medical devices, dentures, implants, prosthetics and more using 3D printing.
Small firms already commercializing devices
Small firms are commercializing 3D-printed medical devices — including surgical instruments, inhalers, smart prosthetic hands, metabolic analyzer masks and more — and can show the path forward. Companies using 3D printing for manufacturing and prototyping can develop new innovative treatments and devices that are customized to patients while remaining affordable.
Medical device company Coalesce Product Development (recently acquired by Novartis) is using 3D printing to develop novel, affordable drug delivery devices such as inhalers and injectors for generic inhalation products that offer significantly better value than brand name alternatives that can cost over $380 per month.
Another company, Restor3d, is bringing the benefits of 3D printing to surgery. Stainless steel instruments traditionally go into more than 132,000 anterior cervical discectomy and fusion procedures performed per year in the U.S. But these instruments had high upfront and ongoing inventory costs and often presented complications in the surgical workflow. Restor3d uses the design freedom and constant improvement enabled by 3D printing to change how spine surgeons operate and improve surgical care delivery. The company’s first 3D-printed, procedure-specific polymer instruments for foot and ankle and cervical spine implants were made from a combination of metal and 3D-printed polymer parts, benefitting hospitals with reduced sterilization and inventory costs.

The 3D-printed Tension Square [Photo courtesy of Formlabs]
These companies and innovators have successfully commercialized 3D-printed end-use parts, as the technology stack is now more accessible than ever. Collaboration within the industry will enable medical device firms to learn from these early examples to chart their own path to commercialization and improve patient outcomes, reduce costs and improve efficiency.
Designing medical devices with 3D printing

Formlabs 3D printers [Photo courtesy of Formlabs]
- Understanding biocompatibility and sterilization requirements of the intended device and reviewing relevant documentation from manufacturers.
- Becoming familiar with quality and regulatory requirements and best practices for leveraging additive manufacturing while maintaining compliance.
- Reviewing 3D printing resources and evaluating various print engines (e.g. FDM, SLS, SLA) and materials with requirements for part performance and manufacturing system compliance in mind.

Gaurav Manchanda is medical market development director at Formlabs. [Photo courtesy of Formlabs]
The opinions expressed in this post are the author’s only and do not necessarily reflect those of MedicalDesignandOutsourcing.com or its employees.