 Lucira
Lucira
The FDA issued an EUA for the first at-home COVID-19 rapid diagnostic test in November.
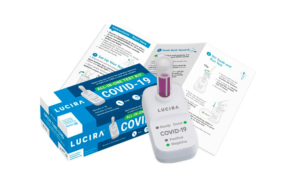
The diagnostic test, developed by Lucira, is an all-in-one prescription test kit — a molecular (real-time loop-mediated amplification reaction) single-use test that detects SARS-CoV-2.
Lucira’s test is authorized for home use with self-collected nasal swabs in people ages 14 and older whom a healthcare provider suspects of having COVID-19. The test was also authorized for point-of-care settings, such as doctor’s offices, hospitals and more, for all ages.
To self-administer the test, users swirl the sample swab in a vial before placing it in the test unit. It’s possible to read the results from the unit’s light-up display in 30 minutes or less to show whether a person is positive or negative for SARS-CoV-2.




