AblaCare seeks a better treatment for PCOS
 Technology: A minimally invasive approach to PCOS-related infertility.
Technology: A minimally invasive approach to PCOS-related infertility.
Unmet need: Find an alternative to infertility treatments with high costs, high risks and side effects.
Polycystic ovary syndrome PCOS is one of the most common female endocrine disorders (up to 10% of women are thought to be affected) and is thought to be the leading cause of female infertility caused by ovulatory dysfunction, accounting for up to 75% of such dysfunction. While effective, current treatments for PCOs are associated with significant risks and side effects: first-line drug-based treatment raises the risk of multiple pregnancies and has common side effects and resistance (up to 40%), second-line surgical ovarian drilling (tissue ablation) has limited clinical adoption due to invasiveness, and third-line IVF is costly and often associated with a higher risk of multiple pregnancies.
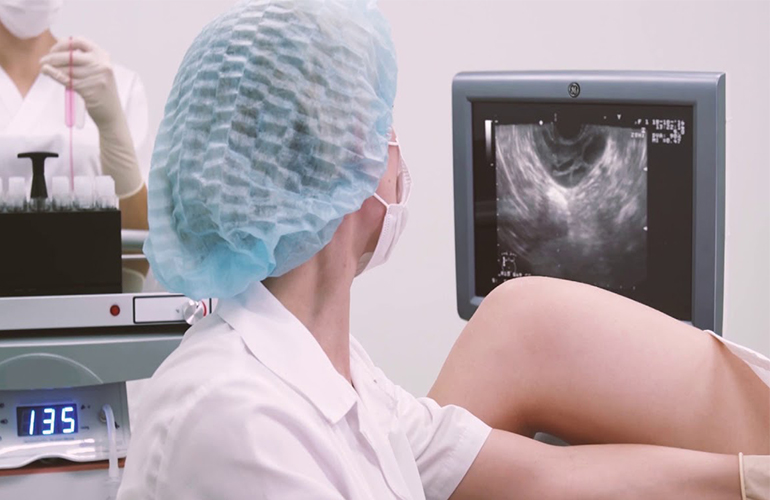
The AblaCare approach would replicate surgical ovarian drilling approach, in a less invasive and less risky manner. Ablacare’s device would comprise a custom radiofrequency generator, and a disposable element (handle, needle, and bipolar electrode catheter deployed through the needle). Once clipped on a standard transvaginal ultrasound probe, the device would be guided to the ovary. The needle would be used to puncture the ovarian capsule, and the bipolar electrode catheter would then be advanced through the needle to perform the ablation in a precise and controlled manner. The electrodes would then be pulled back and re-deployed to perform sequential ablations to achieve the desired ovarian tissue destruction.
The procedure will be simple enough to be performed in the fertility clinic setting by surgeons, reproductive endocrinologists, and eventually general gynecologists.
The company is part of medtech accelerator MD Start. It is currently finalizing and validating the design of its prototypes for pre-clinical and clinical trials.




