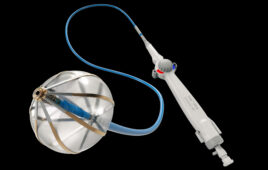
Partnering with a vertically integrated manufacturer can help engineers optimize designs, improve manufacturability and accelerate new product development.
Michael Holt, Integer

(Image courtesy of Integer)
Medtech design engineers face extraordinary opportunities and challenges created by rapidly advancing clinical markets. To quickly bring innovative new products to market, it is important for medical tubing manufacturers to understand thermoplastics, thermosets, filmcast, extrusion, and value-added downstream processes vital to the production sequence.
Electrophysiology catheters, structural heart delivery systems, microcatheters and other tube-based medical devices use a range of polymer tubing constructions and materials, including polyimide, urethanes, nylons, PTFE and PEBAX.
Combining different materials and constructs is vital to achieving the desired balance of strength, flexibility, torque response, kink resistance, and other performance characteristics required in the finished assembly. A vertically integrated manufacturer can work with all of these to improve supply chain coordination and control over inputs.
Here are six critical value-added capabilities that enhance the development process for medical tubing:
Catheter assembly
Assembly of finished tube-based devices requires a variety of services. Contract manufacturers with a full complement of secondary operations, assembly, packaging, and sterilization capabilities provide additional value and supply chain leverage to medical device OEMs and start-ups.
Design for manufacturability (DFM)
The application of DFM as an engineering discipline optimizes the design of complex catheter shafts and other medical tubing assemblies to reduce manufacturing costs. DFM enables correcting potential problems in the design phase, the least expensive point in the development process to address issues.
Reinforced composite tubes
Catheters and other tube-based medical devices requiring high torque response, burst pressure, pushability, steerability and kink resistance typically feature a braid or coil-reinforced shaft design. Multiple variables are at play when designing reinforced shafts. Generally, a reinforced shaft has an inner liner, a braid or coil reinforcement layer(s) and an outer jacket. These elements must be married together to achieve required dimensions while accounting for inherent physical limitations such as liner thickness, outer jacket thickness, and wire density (defined as pic per inch for braid or pitch for coils).
Trade-offs are inherent in designing reinforced shafts. For example, optimizing the device shaft for torque response may result in a braid pattern that is not optimal for flexural stiffness. Modeling these complex reinforced shafts in the virtual space can cut weeks or months off the development timeline.
Reflow shafts
Reflow is a manufacturing process that produces multi-layer shafts by reflowing (or melting) inner and outer jacket materials to construct a composite shaft. Various wall thicknesses, durometers and support structures can be incorporated into a composite shaft to optimize strength, kink-resistance, steerability and torsion control to the shaft design. The reflow process enables all layers of the complex, engineered shaft to be integrated and accommodates a broad range of configurations spanning 2 Fr to 36 Fr.
Secondary operations
Secondary operations can create different tubing features and profiles. These can include hub molding, overmolding, heat treating/annealing, flaring, hole drilling, skiving, pad printing, thermoplastic flattening/shaping forming, tipping, sealing, etching, welding, bonding and swaging. As a precursor to final device assembly, working with a contract manufacturer who provides such secondary operations may cost less than sending components through multiple manufacturers and thus simplify the supply chain.
Virtual tubing model analysis
Conventional medical device prototyping relies on a manual, time-consuming trial-and-error process involving multiple rounds of material procurement, prototype assembly and bench testing. Using virtual tubing model analysis dramatically decreases the time and resources needed for physical prototyping of medical tubing – enabling engineers to model complex, multilayer braided tubes in minutes versus hours, and to quickly and easily predict performance.
Design engineers face numerous pressures to deliver on aggressive development timelines. The right polymer tubing manufacturing partner can enhance innovation and project execution.
Michael Holt is principal applications engineer at Integer (Plano, Texas). Holt has more than 20 years of experience in the design and development of complex catheter-based devices and holds a Bachelor of Science degree in Mechanical Engineering from the University of Tennessee.
The opinions expressed in this blog post are the author’s only and do not necessarily reflect those of Medical Design and Outsourcing or its employees.