Medtronic: Hugo robotic surgery system
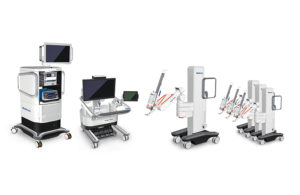
Medtronic’s Hugo robot-assisted surgery system [Image courtesy of Medtronic]
Medtronic management anticipates Hugo’s launch outside the U.S. and Europe, too, with some initial revenue contribution expected to come into play during its fiscal 2021 fourth quarter. The Hugo system is meant to rival Intuitive Surgical’s da Vinci robots.
“We feel really great about our platform, the feedback we’ve got, the open console, the modularity upgrade, ability to leverage of our surgical instrumentation,” Medical Surgical EVP and president Bob White said during a February earnings call.
Other Medtronic devices to watch out for:
- The Stimgenics Differential Target Multiplexed spinal cord stimulation system using Medtronic’s Intellis platform, acquired last year. In January, the company touted three-month results from a large, multicenter, randomized controlled trial. The data showed that 80% of patients with chronic back pain treated with the system reported at least 50% pain relief compared with 51% of patients treated with conventional spinal cord stimulation.
- The Percept PC deep brain stimulation (DBS) system with BrainSense technology senses and records brain signals while delivering DBS therapy to people with neurological disorders such as Parkinson’s disease, essential tremor, primary dystonia, epilepsy and obsessive-compulsive disorder. BrainSense enables more personalized, data-driven neurostimulation treatment, according to Medtronic, which gained the CE mark in the E.U. last year. No word yet from the FDA, which is reviewing the device.
- The MiniMed 780G next-generation insulin pump, which received CE mark approval in Europe in June 2020. The closed-loop insulin pump system is indicated for treating Type 1 diabetes in people aged 7 to 80. It uses Medtronic’s SmartGuard algorithm to automate the delivery of basal insulin and correction boluses every five minutes.




