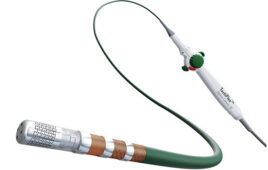
Abbott today announced it has enrolled the first participants in its TactiFlex PAF IDE study to evaluate the performance of the TactiFlex Ablation Catheter, Sensor-Enabled.
Abbott today announced it has enrolled the first participants in its TactiFlex PAF IDE study to evaluate the performance of the TactiFlex Ablation Catheter, Sensor-Enabled.
The catheter is designed to treat people suffering from paroxysmal atrial fibrillation (PAF) with symptoms that are unable to be managed by medication.
TactiFlex combines Abbott’s contact force-sensing technology within the TactiCath SE catheter with a catheter tip design found in the company’s Flexibility irrigated ablation catheter.
Get the full story on our sister site, Medical Tubing + Extrusion.