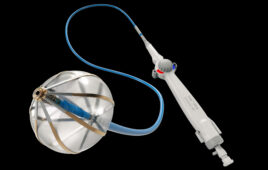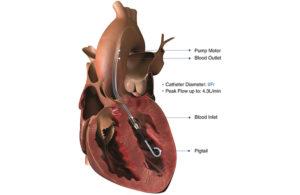
The Abiomed Impella CP heart pump [Image courtesy of National Institutes of Health]
Data from the PROTECT III FDA post-approval study of Impella 2.5 and Impella CP in high-risk percutaneous coronary intervention (PCI) patients showed reduced rates of MACCE — a composite of death, stroke, myocardial infarction and repeat procedures.
PROTECT III builds on the PROTECT II randomized controlled trial (RCT) which found, when compared to intra-aortic balloon pump (IABP), Impella use led to a 29% reduction in MACCE at 90 days, the company said.