Pericytes are the primary locus of matrix-mellaproteinase-9-dependent (MMP-9) capillary damage and blood leakage during ischemia, according to preclinical findings reported by Medical University of South Carolina (MUSC) investigators in an article published online on November 14, 2016 by The Journal of Neuroscience. In vivo two-photon microscopy revealed MMP-9 activity and plasma leakage disproportionately occurred at locations where pericyte somata were attached to the endothelium. These results suggest that pericytes, normally essential for blood-brain barrier (BBB) function, contribute to capillary damage during stroke.
The BBB–a highly specialized vascular structure–prevents the entry of blood-borne substances that can harm the brain (e.g., neurotransmitters such as glutamate, clotting factors such as fibrin, and free radical-generating substances such as iron). During ischemic stroke and related cerebrovascular diseases, the BBB is damaged, allowing incursion of blood plasma that injures neurons and other structures essential for normal cerebral function.
The role of pericytes as builders and custodians of the BBB is well recognized, but how pericytes respond to blood flow loss in the adult brain has largely been a mystery, until now.
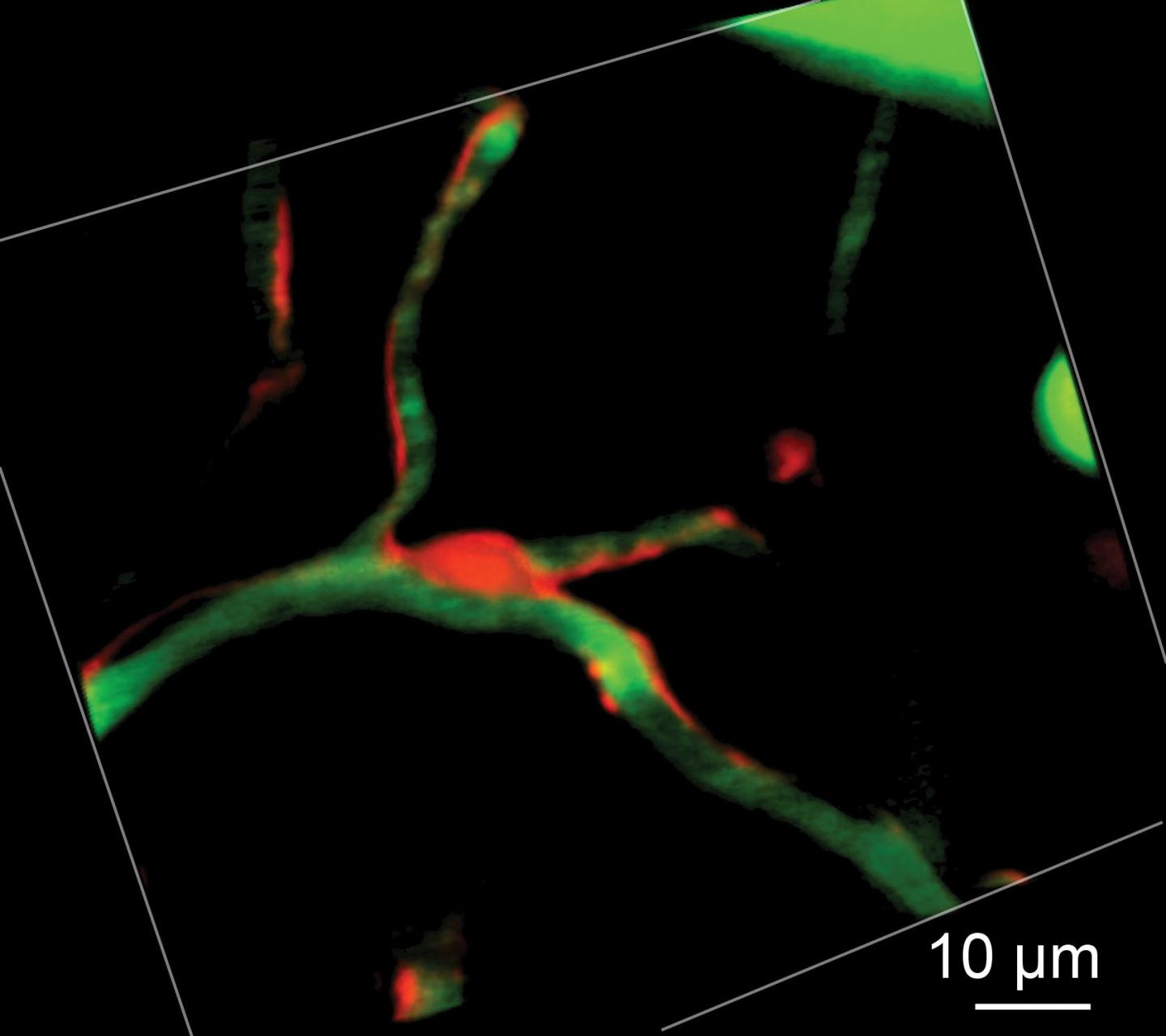
This is a fluorescent pericyte cell body (red) with processes extending along adjacent capillaries (green). (Credit: Andy Y. Shih, Ph.D., and Robert Underly, Medical University of South Carolina)
MUSC researchers recently harnessed cutting-edge technology to image pericytes in the intact brains of live mice and to spatially and temporally track the proteolytic enzyme, MMP-9, as the BBB degraded and blood began leaking into the brain.
Findings from this novel study not only provide critical new information about pericytes as a potent source of MMP-9 during BBB leakage but also open new discovery pathways for future therapies in neurological conditions involving ischemia.
It all began when Robert Underly, a Ph.D .candidate in the MUSC College of Graduate Studies Neuroscience Program and first author on the article, noticed that, when a laser was used to induce ischemic strokes in the laboratory, BBB leakage occurred at very specific sites along the capillaries.
“I’d assumed that blood leakage occurred along the entire capillary length,” said Underly. “But it wasn’t like that. There were hot spots that leaked first and more than the rest of the capillary bed. That was really unexpected.”
Andy Y. Shih, Ph.D., Assistant Professor of Neurosciences and senior author on the article, and his team followed up on Underly’s observation. “We found a very close association between where the round cell bodies of pericytes were located and where the leaks occurred,” said Shih. “So that was our first clue that the pericytes were possibly doing something harmful in the early stages of an ischemic stroke.”
It is well known that the BBB becomes dysfunctional when genetic defects disrupt pericyte-endothelial signaling from birth. However, very little is known about how normally developed pericytes in the adult brain respond during acute ischemia, and only one or two studies have investigated this in vivo.
“Pericytes have a lot of potential functions–they seem to be a sort of a jack-of-all-trades,” said Shih. “We’ve had an idea of what these cells do, but we haven’t been able to visualize and track them in vivo until recently.”
The team was also intrigued by a handful of published studies showing that various inflammatory signaling cascades can induce pericyte MMP-9 expression.
“The problem is that, like pericytes, MMPs are hard to study in vivo–most of what we know about them is from studies on cultured cells or extracted brains,” said Underly. “We wanted to probe this process in live animals so we could see the spatiotemporal relationship between pericytes and MMP activation in vivo–in the acute stroke time frame.”
To do this, the team combined several novel tools to create a unique study protocol using transgenic mice, two-photon fluorescent microscopy, and a fluorescent gelatin-based reporter of MMPs that only one other research group had ever used to study the intact brain.
The researchers also used photothrombosis to block blood flow in a small area of the capillary bed and imaged transgenic mouse lines expressing bright fluorescent reporters specifically in the pericytes to clearly identify them.
“The successful combination of technologies is certainly one of the innovations of this project,” said Shih. “It’s the first study to combine these tools to look at the relationship among pericytes, MMP activity, and BBB leakage in ischemia.”
Their findings revealed that ischemia resulted in extremely rapid (within tens of minutes), localized activation of MMP-9 and plasma leakage. Furthermore, plasma leakage occurred preferentially where the pericyte somata adjoined the capillary wall–not homogeneously along the length of the capillaries as previously imagined by the group.
These results provide strong evidence that pericytes–normally protectors of the BBB–contribute to early BBB degradation during ischemic stroke.
Using the new technology, the team did not have to extract and cut the brain and so did not lose the structure of the vasculature and blood flow.
“We had an intact system and could see where things were coming from and we were very surprised,” said Shih. “I thought, ‘I’ve been looking at this for years and I never knew that there was this beautiful co-localization.’ It told us we were looking at something really interesting. The pericytes seem to nurture or damage the BBB depending on the conditions they’re put in.”
This discovery opens many directions for further study and could eventually lead to new therapeutic options for patients experiencing an acute stroke.
“The very rapid reaction we saw to ischemia is really important and provides clues to potential mechanisms by which MMPs may be up-regulated,” explained Underly. “This is a future direction for our research–to define upstream regulators of this process that can be therapeutically targeted.”
“The findings raise so many new research questions,” said Shih. “For example, why do pericytes have so much MMP? What are they doing with it? What happens to pericytes days to weeks after an ischemic event? There’s so much still to be understood about the acute phenomena–we’re focused on that for now. In the future, we could look at later, post-injury, events to see what happens next in the life of the pericyte.”
Indeed, in vivo cellular-level imaging research has a bright future.
“There’s a renaissance happening with the development of new molecular tools to image and modify cell function in vivo,” said Shih. “We’re going to see a lot more integration between tool-makers and in vivo imaging groups in the next decade or two. There are going to be many more studies looking at the intact brain.”
“It’s important to fund projects like this because with in vivo imaging we’re able to track exactly what happens when brain function breaks down,” said Underly. “The disease state occurs in front of our eyes.”




