Editor’s Note: This article was written by Chris Krampitz, Strategy and Innovation for AM Technologies, UL
Editor’s Note: This article was written by Chris Krampitz, Strategy and Innovation for AM Technologies, UL
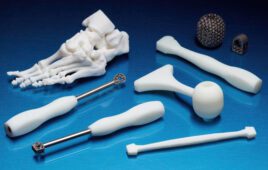
Additive manufacturing (AM) is a complex process when it comes to compliance. It is a specialized process with a lot more sources of variability than traditionally manufactured devices. This variability comes at every stage of development: design, materials selection, processing, and post processing. And those sources of variability are less understood than traditionally manufactured devices. As such, there are singular challenges with compliance to make sure those devices are manufactured consistently against the regulatory requirements, industry standards, and even the customer specified requirements.
Another factor that complicates the compliance of 3D printed products is that the test methods for assessing the performance of the devices or implants are used a bit differently, or applied differently, than in traditional manufacturing. For example, nondestructive techniques, like CT scanning or ultrasonic scanning have been applied to traditionally manufactured devices and additive. AM manufacturers and compliance bodies are still testing to see limitations of nondestructive tests as well as when those tests are suitable and when they’re not. That adds a bit more complexity in assessing compliance.
A major factor to understanding the complexity of 3D printing is to consider that the prime benefit of the process is also the greatest difficulty in terms of compliance. It seems obvious, but the design changes can put devices out of compliance without the designer fully understanding that ramifications. In terms of compliance, change is not good, and variability is bad. In traditional manufacturing the rule is that you have to reduce variability; but in AM the variability is the source of benefit so we have to find ways of embracing that variability and learning how to harness and control it and direct it.
FDA Guidance
In order to harness those benefits and mitigate the risks of variability, FDA has released a guidance document Technical Considerations for Additive Manufactured Devices. There is quite a lot in the document but there are some important that can be highlighted. The guidance is a major step forward in adding clarity to manufacturers of medical devices that are trying to use the technology.
The purpose of the guidance is to outline the technical considerations associated with additive manufacturing processes, provide recommendations for testing and characterization of the devices, as well as factors that occur during the development stages. This is for devices manufactured exclusively with addititve processes as well as devices in which AM is used in one or more steps. Devices don’t necessarily have to be entirely produced with additive manufacturing.
The guidance is broken up into two major sections. The first section is associated with design and manufacturing considerations. FDA focused on providing recommendations around controlling the designs, and the different aspects of the manufacturing process. It assesses the different steps that need to be considered, and what happens during those steps.
The second topic area, the device testing considerations, provides information on the type of data that should be submitted during 510(k) or the pre-market approval applications. It helps a device manufacturer, device designer, that’s developing these devices figure out what information they should be submitting.
The document tries to answer, “How do I need to think about the design and manufacturing of these medical devices, and what type of information I need to submit for the FDA to even review the applications?” Manufacturers should be thinking about that right now, and that’s the number one priority.
One thing the guidance does not do is address the point-of-care device manufacturing. If you’re trying to manufacture at a hospital or specific location, this guidance doesn’t really talk about those situations. It also doesn’t address any biological, cellular, or tissue-based production using the technology, like bioprinting.
With manufacturing at point-of-care and bioprinting, there is still a lot more in research and development to be done. As those options mature more, we’ll start seeing those ideas incorporated into the guidelines. I expect point-of-care manufacturing to happen sooner than bioprinting, because there’s still quite a bit of research and development work that needs to be done on bioprinting.
Embracing Variability
Medical manufacturers should understand that the process to design and manufacture these medical devices using the technology is quite complicated, and it requires a holistic supply chain view. Partnering with a service provider can help through the entire process. There are variables in the manufacturing process that need to be examined, which don’t correlate with decision-making processes in traditional manufacturing. Topics such as software workflow, imaging resolution of patient scans that transferred into CAD files have not been part of traditional manufacturing, but they are a significant part of the benefits of AM technology.
But those benefits also add complexity, and having a partner that understands the variability is important to successfully move from development through production, and achieve FDA approvals.