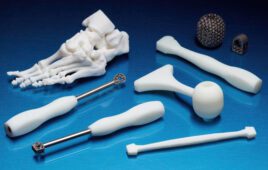
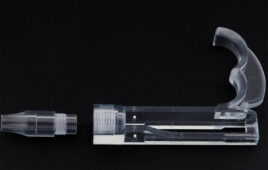
Becton Dickinson has begun using Carbon’s 3D printing technology to manufacture its hemocytometer adapter.
Becton, Dickinson and Company (BD) has begun producing parts based on Carbon’s digital light synthesis technology, including BD’s first production part using additive manufacturing — a hemocytometer adapter for the company’s Rhapsody single-cell genomic analysis system.
This component would have been impractical and expensive to injection mold, according to Redwood City, Calif.–based Carbon, which said it offered BD a cost-effective solution to address difficult-to-mold geometries without sacrificing part quality.
BD had identified products struggling with part geometries or economies of scale with both injection molding and milling and began a program to examine additive processes, materials, and companies claiming to meet demanding medical applications. Carbon produced a case study examining how BD identified the application for additive manufacturing and why it selected Carbon and its MPU 100 medical-grade material for production. MPU 100 is Carbon’s first material designed to help medical product manufacturers accelerate product development cycles and improve the quality of their products. Similar to Carbon’s rigid polyurethane materials, MPU 100 is a two-component material that produces isotropic parts with fine features.
The BD genomics team was building a single-cell genomic analysis system that makes it possible to understand cellular form and function on the basis of individual cells. Traditional assays such as microarrays and bulk RNA sequencing, which take an average of measurements across multiple cells, hide subtle differences between individual cells.
The BD Rhapsody system was designed to overcome this limitation and make it possible to identify and characterize rare cell types, allowing researchers to understand biological processes in fields ranging from immunology to oncology. A critical component of this product is the hemocytometer adapter, which integrates a fluidic microwell component into an optical system. The fabricated holder must accommodate the dimensions and operational requirements of both a fluidic “slide” and the existing imaging technology.
By transitioning this part to a digital manufacturing process, BD could iterate at will, integrating feedback from Carbon to improve its product and optimize the manufacturing process to increase print speed by 55% and reduce resin usage by 7%. The process also accelerated BD’s product development cycle, and economically produced final parts, Carbon said.
“We worked closely with the Carbon team to leverage the biocompatibility and sterilization data already generated for MPU 100 material,” said Larry Monahan, manager of BD’s advanced prototyping, corporate computer-aided engineering (CAE) team, in a statement. “Then we partnered with BD’s toxicology group to generate our own internal data.”
Read more about the case study here.