Edwards Lifesciences: +36%
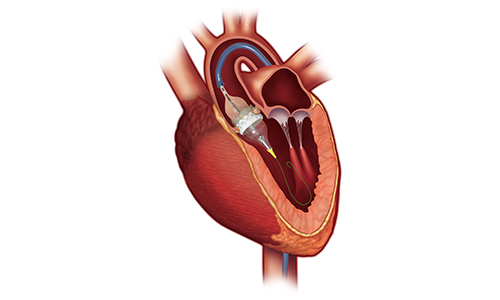
Edwards Lifescience’s Sapien 3 TAVR [Image courtesy of Edwards]
Irvine, Calif.–based Edwards touted it as the first commercially cleared transcath therapy for treating tricuspid heart valve disease. The company acquired the Cardioband tech when it bought Valtech Cardio in a deal worth $690 million, which closed last January.
In May, the company inked a deal to repurchase an aggregate of $400 million in its common shares.
The company continued to battle Boston Scientific (NYSE:BSX) in a global tussle over transcatheter aortic valve replacement patents. In October, Boston won an injunction barring Edwards’ the Sapien 3 Ultra valve from the German market, after the District Court of Düsseldorf found that Edwards infringed a patent Boston acquired with the $435 million purchase of Swiss valve maker Symetis in May 2017. The infringement concerns the fabric used on the valve seal, Marlborough, Mass.-based Boston Scientific said.
In October, Edwards said that it launched a U.S.-based pivotal trial of its self-expanding Centera transcatheter aortic valve exploring its use in treating severe symptomatic aortic stenosis in intermediate risk patients. The Centera valve is designed to be repositionable and retrievable, and is delivered through the use of a 14 French motorized delivery system in which the valve is pre-attached to the mechanism for quick preparation, Edwards said.
Among the large legacy medical device companies, Edwards is a big R&D spender. During its most recent fiscal year, it put nearly $553 million into R&D, an amount that represented more than 16% of its revenue.
The FDA approved Edwards Lifesciences to market its Sapien 3 Ultra transcatheter aortic valve replacement device in the U.S. The device was approved for severe, symptomatic aortic stenosis patients who are at greater risk of complications during open heart surgery.







