ImageIQ, an Imaging Software and Contract Research Organization focused on supporting preclinical research and clinical trials, announced that it has successfully supported Biogennix in completing its recent long-bone fracture healing and spine fusion preclinical studies. Both of these studies generated data that resulted in 510(k) marketing approvals by the US Food and Drug Administration (FDA).
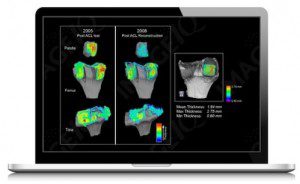 Biogennix products are indicated for voids or gaps in the extremities, posterolateral spine and pelvis that are not intrinsic to the stability of the bony structure. The product provides bone void filler that resorbs and is replaced with bone during the healing process. Biogennix selected ImageIQ to support these studies because of ImageIQ’s history of leveraging its 2D and 3D imaging expertise, combined with its unique approach to software-based quantitative image analysis, to generate imaging data that is more quantitative and objective than traditional approaches. Specifically, ImageIQ provided histology and micro-computed tomography quantitative imaging and analysis to strengthen the Biogennix 510(k) submissions, and provided data for a more objective and quantitative product efficacy claim.
Biogennix products are indicated for voids or gaps in the extremities, posterolateral spine and pelvis that are not intrinsic to the stability of the bony structure. The product provides bone void filler that resorbs and is replaced with bone during the healing process. Biogennix selected ImageIQ to support these studies because of ImageIQ’s history of leveraging its 2D and 3D imaging expertise, combined with its unique approach to software-based quantitative image analysis, to generate imaging data that is more quantitative and objective than traditional approaches. Specifically, ImageIQ provided histology and micro-computed tomography quantitative imaging and analysis to strengthen the Biogennix 510(k) submissions, and provided data for a more objective and quantitative product efficacy claim.
ImageIQ
image-iq.com
Biogennix
biogennix.com




