A Minnesota judge rules that federal law prevents patients from claiming damages against medical device giant Medtronic, just as an Illinois judges rules that a similar case may proceed.
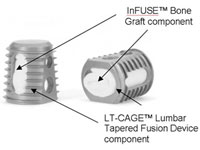
The battle over Medtronic’s (NYSE:MDT) Infuse bone-growth protein continues to be a roller-coaster, with judges in 2 states issuing seemingly conflicting rulings about the validity of patient injury lawsuits against the Minnesota medical device giant and its troubled Infuse bone-growth protein.
With the dismissal of 1 lawsuit while another is allowed to proceed, patients and medical device makers alike are looking for potential weaknesses in federal preemption laws, which have protected many medical device makers by effectively squashing liability lawsuits against products approved by the FDA.




