Many factors — including the clinical phase, length of study and number of sites — impact overall trial costs. However, many organizations are moving forward with these strategies in hopes of lowering overall spends.
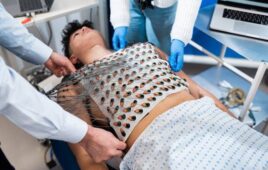
Many life science companies look to risk-based monitoring (RBM) as a way to reduce the costs associated with clinical studies. Whereas on-site monitoring is a significant expense for clinical trials, RBM approaches allow monitors to communicate remotely with well-adjusted clinical sites rather than using valuable man-hours traveling for on-site visits.
According to primary intelligence provider Cutting Edge Information, RBM monitors also review only a percentage of source data to verify accuracy, further reducing the time needed to validate site data. Reducing the time necessary to work with each site often means that teams need to employ fewer site monitors or clinical research associates (CRAs) per study, greatly impacting the overall potential trial costs.
Risk-Based Monitoring: Inject Remote Risk Assessment to Optimize Clinical Trial Outcomes by Cutting Edge Information provides benchmarks on budgets and staffing for risk assessment teams and RBM activities. The report also includes best practices for implementing RBM strategy and tactics associated with risk-based monitoring usage. The report segments companies’ existing use of risk-based monitoring strategy by therapy and trial phase to showcase RBM’s role in efficient clinical trial management.
The study will help clinical operations executives:
- Avoid implementing overly complicated RBM techniques
- Balance existing budget and staffing resources
- Map out clinical strategy well before trial initialization
Cutting Edge Information
cuttingedgeinfo.com