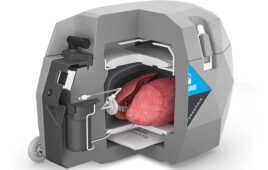
In a coronavirus pandemic in which patients on the best commercial ventilators will die, are garage enthusiasts ready to stand behind their work?
Scott Thielman, Product Creation Studio

There are presently serious efforts underway at universities to find out ways to assemble existing equipment into ventilators, including at MIT (bottom left), the University of Minnesota (bottom right), and a hackathon team called OpenVent (upper right).
Almost a decade ago, Dr. Robert Clark, a pulmonologist then at Regional Hospital for Respiratory and Complex Care near Seattle, approached my company with a warning and an opportunity.
To paraphrase, he said, “There aren’t enough critical care ventilators and there isn’t enough oxygen when the next pandemic comes. I want to build a product to solve both deficiencies and meet the guidelines I helped establish with the federal government on price and performance. Can you help?”
So began my crash course in ventilator modes, oxygen concentration and breathing circuits. A tour of the critical care pulmonology wing left an indelible memory of sedated, intubated patients. Dead, but for the whirring, flashing machines they were connected to and the constant attention of the respiratory therapists. Clark explained that patients with acute respiratory distress syndrome (ARDS) may need support for months as their lungs recover. And weaning them off ventilation could be the most dangerous time of all; many can’t regain the strength to breathe on their own.
All of this shocked us into action. We assembled a scrappy team to design the WynVent, a combination ventilator/concentrator that would save lives when there were too many of these patients and not enough of the magic machines to keep them alive. On top of it all, they needed to be affordable. Clark said that critical care ventilator prices had roughly paralleled the cost of a luxury Mercedes Benz sedan over the years; the WynVent needed to be lower-cost to allow mass production and bulk buys. It was a laudable goal and drove our team to work overtime, our owners to invest, and me to lose sleep dreaming of inventions to add reliability and remove cost.
Over the course of the next year we built prototypes, ventilated test lungs and agonized over how to keep zeolite (concentrator material) dry. In our case, there was never enough oxygen to hit the target level for a truly compromised ARDS patient. We pushed vendors and academics for ways to compress more air, remove weight from valves and ease the software development burden. The WynVent was our moon shot.
As device development professionals, we managed our design inputs against one of the longest lists of standards I have seen before or since (see the current FDA emergency use authorization for a sampling of these). We built our risk matrix, started building a test plan and had an initial meeting with the FDA to discuss the ventilator.
A complication was that respiratory therapy is not a rote, homogenous type of treatment. Respiratory therapists employ numerous strategies and protocols and many types of equipment to treat myriad patient conditions; each logical branch leads to outcomes with risks that must be evaluated and mitigated. These weren’t pandemic times, but even getting to that meeting was a year of work for a team of about six people.
As with many startups, the funding became increasingly intermittent and life intervened to effectively mothball the WynVent. But the experience informs my perspective on today’s race to build new ventilator products in record time. While I want to go “all in” again on a project to save lives, I’m realistic about what it takes to bring a new ventilator to market.
Regulatory bodies may ease compliance burdens, but there is an implied fitness for purpose when you build and deliver a product. When the stakes are high for product failures, the governing bodies tend to ask for organized ways to demonstrate safety; thus, standards are born. The list of standards that affect modern mechanical ventilators is not long because regulators are evil. It is long because the credible ways to injure are many for a product intended to keep us alive.
I love the can-do attitude and Apollo 13 references I see online. That inventive optimism is what excites me about engineering and forms a big part of the work we do at Product Creation Studio. But keep in mind that when a tool leaves your garage to be pressed into emergency service, it is easy for enthusiasm overshadow concern about fitness for purpose; it’s on you if it fails. This is why we see requests for collaboration and help on testing from some of the early-stage projects.
In a healthcare scenario where many patients on the best commercial equipment will die during treatment, are the garage enthusiasts really ready to stand behind their inventions? I can’t answer for you, but I would not want to skip many of the tests to show that requirements have been met (verification) and even fewer of the tests to show that the product meets the intended user needs (validation).
If you are a DIY inventor working on a novel respiratory solution, I urge you to adjust your sights toward a goal that may have real impact: affordable ventilator solutions available in the coming years, developed at a pace that is commensurate with the gravity of care being provided. These innovations may drive cost down and accessibility up for mechanical ventilation in the future. Maybe they won’t save lives in New York next week, but they could save lives in Kenya in a year or two.
For the groups looking to press existing medical devices into service as ventilators, keep working your long nights because your product could be launched much sooner. FDA has made an emergency use authorization pathway for you and there is a reasonable default to a standard of care (manual resuscitation bags) that most healthcare workers recognize. Building on a well-designed and tested device should allow you to partition and shorten your risk analysis and test burden. Use information made available by groups like Medtronic, which shared complete device specifications for a commercial ventilator, to guide you on which risks, requirements and documentation are crucial.
Realistically, the current pandemic surge is a procurement and manufacturing challenge. New products just won’t be available during the looming peak need whereas existing products and production lines can respond. This is the current moonshot and it should be all-hands-on-deck to accomplish it.
Scott Thielman is CTO and co-founder of Product Creation Studio. Thielman has participated in all aspects of product development for clients including Philips, Johnson & Johnson, and Lilly. He holds a Ph.D. in mechanical engineering from the University of Washington and is a licensed professional engineer.
The opinions expressed in this blog post are the author’s only and do not necessarily reflect those of Medical Design and Outsourcing or its employees.