
[Photo by Nicole Harrington on Unsplash]
Thousands of medical device industry insiders, many based in the area, attend the show. Strolling around the show floor at the Minneapolis Convention Center, one gains an appreciation of the strength and depth of the contract manufacturing and outsourcing ecosystem supporting the major medical device hub based around Minneapolis-St. Paul.
Here are some of the exhibitor technologies highlighted at this year’s show:
Excelitas Technologies
Booth #1432
Excelitas Technologies (Waltham, Mass.) at MD&M Minneapolis plans to tout its broad offering of OmniCure LED UV curing systems. Products on display include:
- The OmniCure LX500 LED spot UV curing system, which features up to four LED heads with up to 16 W/cm2 peak irradiance and proprietary feedback technology. OmniCure LX500 is ideal for use in medical devices such as catheters, cannulas, endoscopes and syringes, according to Excelitas.
- The OmniCure S2000 spot UV curing lamp system, which Excelitas says is ideal for precision adhesive curing in a variety of industries, particularly medical device bonding applications.
- The OmniCure AC8-HD (high dose) LED UV curing system, which offers higher optical power and enhanced performance in the same air-cooled mechanical enclosure as their AC8 and AC9 predecessors. The light sources, according to Excelitas, are ideal for large area curing in a variety of medical applications.
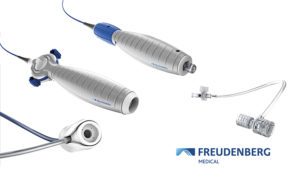
[Image courtesy of Freudenberg Medical]
Freudenberg Medical
Booth #1616
Freudenberg Medical is using MD&M Minneapolis to show off two new catheter handle platforms and a new hemostasis valve. Products touted by the Carpinteria, Calif.–based contract manufacturer will include:
- The Composer Epic catheter handle platform includes what the company describes as next-generation design options. There’s a through lumen, modular distal end and a variety of shaft options for electrophysiology guide, diagnostic and therapeutic catheters.
- The Composer Toccata catheter handle platform is meant to control smaller catheter applications up to 9.5 Fr OD. The platform provides extensive design flexibility with a variety of shaft options, a proximal luer connection and an adjustable tensioning system, according to Freudenberg.
- The HyperSeal Mini is the sixth and latest member of the Freudenberg Medical hemostasis valve family. It brings the automatically adjusting HyperSeal valve technology into 12Fr and smaller applications including integration into the new Composer Epic and Toccata handles.
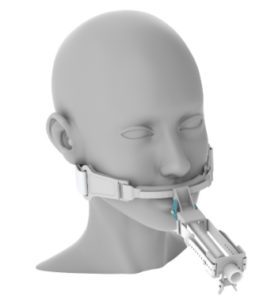
The TubeTrac [Image courtesy of Airway Innovations and Keystone Solutions Group]
Keystone Solutions Group
Booth #1723
Keystone Solutions Group (Kalamazoo, Mich.) is highlighting Airway Innovations’ TubeTrac, a single-use endotracheal tube holder meant to prevent patients from removing their own breathing tube. Keystone is the development and manufacturing partner for the device.
The TubeTrac attaches to the endotracheal tube with a universal clip. Airway Innovations and Keystone say the design makes it nearly impossible for a patient to pull out a tube, and it prevents accidental tube removal. TubeTrac requires two to three times more force to remove the tube compared to the competition, according to the companies. It’s skin-friendly and adjustable.
Design challenges included significant testing and materials selection to ensure the product surpassed all competitive products in strength and safety, Jim Medsker, Keystone Solutions Group’s president, told Medical Design & Outsourcing.
Airway Innovations’ CEO is Eric VanMiddendorp. who is also a biomedical engineer at Spectrum Health Innovations in western Michigan.

Junkosha Multi-Filar cable for catheters [Image courtesy of Junkosha]
Junkosha
Booth #1115
Junkosha will use MD&M Minneapolis next week to launch its Multi-Filar fine wire and cable for catheters. The new technology could enable smaller, more flexible and higher-data-capacity catheters. The new cables have a capacity of up to 60 power lines depending on AWG size or pitch, according to Junkosha. The company designed them to provide not only increased functionality but also more signals into areas of the body that were formerly inaccessible. Read more on our sister site Medical Tubing + Extrusion.

[Image courtesy of Meddux]
Meddux
Booth 2539
Meddux Development Corp. — an engineering design and development firm for single-use interventional and minimally invasive surgical devices — has expanded its finished goods manufacturing capacity. In the past year, Meddux has invested in manufacturing capacity, doubling the size of its Controlled Environment (CE) and adding what the Boulder, Colo.–based company describes as a significant amount of warehouse space. Meddux officials say the company now has the ability to support a medical device company’s entire product lifecycle from concept through commercialization.

[Image courtesy of Medline]
Medline Industries
Booth #1046
At MD&M Minneapolis, Medline is touting its custom packaging capabilities, components, and drapes and gowns. The company creates its custom packs with customer input so that they include the most frequently used items. Standard packs are stocked and ready, but Medline can also include any item neded for a procedure in one of its custom packs. Medline boasts that it keeps the cust of custom packs low through the high volume.
Meier Tool & Engineering and Cretex Medical
Booth #2005
Meier Tool & Engineering (Anoka, Minn.) — part of Cretex Medical — plans to tout its new prototyping services, including RapidStamp capabilities that offer a quick turn of two weeks or less. Meier boasts tool designers and engineers to provide design for manufacturability input and testing at both the high and low range of dimensions and tolerances. The company’s Dedicated Prototyping Center offers prototype tool build, feasibility studies with stamped parts, low volume manufacturing support, and stock materials for prototyping, and a dedicated team with surge capacity.

[Image courtesy of Micro]
Micro
Booth #1119
Micro (Somerset, N.J.) is touting its Precision Electrochemical Machining (PEM) process. PEM removes metal from the work piece by electrochemically dissolving specific quantities of material using predetermined process parameters. Micro specializes in custom tooling featuring multi-part or multi-cavity machining, “contact free” processing and no tool wear. Micro officials boast that their company can produce accurately machined, intricate part with tolerances capabilities of 5 microns (.0002 in.) — perfect for precision orthopedic, surgical components, and more.

Nelipak medical trays [Image from Nelipak]
Nelipak
Booth #1437
Nelipak is touting its design, development and manufacturing capabilities at MD&M Minneapolis. The company specializes in packaging needs for stringent healthcare environments.
The company produces a number of products, including medical trays, procedure trays, sealing machines and protective packaging.

OmniVision’s OV6948 (below) next to a peppercorn. [Image courtesy of OmniVision]
OmniVision Technologies
Booth #1248
OmniVision Technologies (Santa Clara, Calif.) boasts that its OV6948 is the winner of the Guinness World Record for “the smallest image sensor commercially available” with its size of 0.575 mm by 0.575 mm. The company is also announcing the OVM6948 CameraCubeChip — derived from the ultra-small OV6948 to create a fully packaged, wafer-level camera module measuring 0.65 mm by 0.65mm, with a z-height of just 1.158 mm. OmniVision officials say their new tiny medical imagers address market demand for decreased invasiveness and deeper anatomical access. The imagers also address the many challenges posed by reusable medical imaging equipment, including cross-contamination risks and inefficiencies due to high maintenance costs, according to the company.

Qosina boasts that it carries more than 40 different configurations of tuohy borst adapters. [Image courtesy of Qosina]
Qosina
Booth #1816
Qosina (Ronkonkoma, N.Y.), is using MD&M Minneapolis to showcase its tuohy borst adapters. A three-piece assembly comprised of a body, gasket and cap, a tuohy borst adapter is commonly used to prevent the backflow of fluid and facilitate catheter introduction. The silicone valve and cap torque around a tube or instrument to hold it in place. Qosina boasts that it carries more than 40 different configurations of tuohy borst adapters in sizes ranging from 0–18 Fr.
TM Robotics
Booth #723
Visitors to TM Robotics‘ booth at MD&M Minneapolis will be able to check out the TVM 6-axis robot unload and inspect the output of a plastic injection molding machine with highly-accurate synchronicity. TM Robotics (Elk Grove Village, Ill.) boasts that the TVM 6-axis robot is a cost-effective, vertically articulated robot with a lightweight, reliable design.
TM Robotics’ Minnesota and Wisconsin distributor, Werner Electric, which also show off the TVL500 6-axis, THL300 SCARA machine and THL700 SCARA at Booth #805.
This story originally ran Oct. 16. Updated with additional items.







