SpineGuard, a company that develops and markets disposable medical devices to simplify surgeries, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its new PediGuard Threaded DSG device.The device with DSG, i.e. Dynamic Surgical Guidance technology, embedded inside may be used in open or minimally invasive approaches for pedicle screw insertion. It is available in various designs to accommodate surgeon preferences and patient anatomy.
A single-use DSG pin embedded with the bipolar sensor is inserted into the cannula of the threaded shaft and connected to the electronic processor inside the single-use DSG handle. The distal tip of the threaded shaft includes an awl-like tip to facilitate redirection of the device until the tip is past the pedicle isthmus.
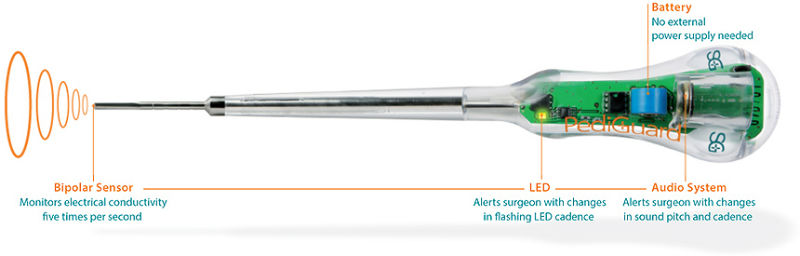
More than 45,000 surgical procedures have been performed worldwide with PediGuard, according to the company. (Photos courtesy of SpineGuard, www.spineguard.com)
For surgeons, the probe reduces steps in fusion surgery. It was introduced in Europe and Asia earlier this year.
SpineGuard, with offices in San Francisco and Paris, was co-founded in 2009 with a goal to bring real-time digital technology into the operating room. The company hopes to establish its proprietary DSG technology as a global standard in surgical care, beginning with safer screw placement in spine surgery and then in other surgeries.
The company’s PediGuard handheld device is capable of alerting surgeons to potential pedicular or vertebral breaches. More than 45,000 surgical procedures have been performed worldwide with PediGuard, according to the company.




