Federal healthcare regulators review Edwards Lifesciences request for an extension to patents for its Sapien transcatheter aortic valve implantation system, the only such device on the U.S. market.
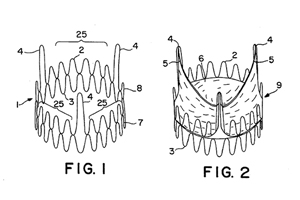
Medical device maker Edwards Lifesciences (NYSE:EW) is seeking an extension for patents related to its Sapien transcatheter aortic valve implantation technology, according to an FDA notice.
Federal healthcare regulators determined the “regulatory review period” that Edwards may use to request its patent extension to amount to 2,473 days, comprising 2,106 days of testing and 367 days of review. Only a portion of that time may be recouped, according to the FDA.




