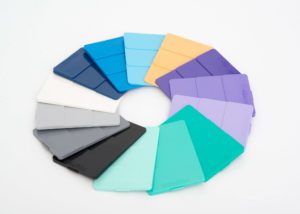
Foster Corporation, a producer of custom polymers for medical devices, introduced a new line of color concentrates using FDA 21CFR 73 subpart D pigments for medical device applications. In support of medical customers in early stages of product development, Foster’s MediBatch color concentrates will be available in single pound quantities.
Medical devices often use custom colored components for branding, aesthetics or functional purposes. Color concentrates, blended with unpigmented polymers are widely used as an economical alternative to pre-colored formulations for injection molded or extruded plastic components. Pigments used for device components that contact the human body, such as catheter tubing, require documentation to be submitted as part of device approval by the FDA. Use of pigments that have been previously evaluated for use in medical devices may reduce the FDA review time for a new device and expedite the approval process.
MediBatch color concentrates are formulated using FDA 21CFR 73 Subpart D listed pigments and a polymer carrier that is compatible with a wide range of base resins, including nylons and polyurethanes. Twelve standard concentrates are available from stock in 1, 5 and 10 pound quantities. Custom colored concentrates are also available on a made to order basis.
Foster
fostercomp.com