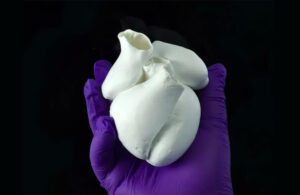
A biohybrid model of a four-chambered heart engineered with focused rotary jet spinning (FRJS) technology [Image courtesy of Harvard SEAS]
They created ventricle structures with the method and then seeded them with rat cardiomyocyte or human stem cell-derived cardiomyocyte cells, according to a news release from the Wyss Institute for Biologically Inspired Engineering at Harvard University. Roughly a week later, they had several thin layers of beating tissue covering the scaffold. The cells in the beating tissue followed the same helical alignment as the fibers underneath.
The bioengineers, who were from the Wyss Institue and the Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS), were then able to run experiments that compared the performance of ventricles made from helically aligned fibers with those made from circumferentially aligned fibers. They found the helically aligned tissue outperformed the circumferentially aligned tissue on every front. The result provided evidence backing a decades-old hypothesis — biomathematician Edward Sallin’s idea from 1969 that the heart’s helical alignment is critical to achieving large ejection fractions.
“In this case, we went back to address a never tested observation about the helical structure of the laminar architecture of the heart. Fortunately, Professor Sallin published a theoretical prediction more than a half century ago, and we were able to build a new manufacturing platform that enabled us to test his hypothesis and address this centuries-old question,” said Kit Parker, professor of bioengineering and applied physics at SEAS, an associate faculty member at the Wyss Institute and senior author of the research paper appearing in Science.
Developed at SEAS by Parker’s Disease Biophysics Group, focused rotary jet spinning (FRJS) can provide high-throughput fabrication of specifically aligned fibers. Diameters can range from several micrometers to hundreds of nanometers.
“The human heart actually has multiple layers of helically aligned muscles with different angles of alignment. With FRJS, we can recreate those complex structures in a really precise way, forming single and even four chambered ventricle structures,” said Huibin Chang, a SEAS postdoctoral fellow and co-first author of the paper.
Here’s how the Wyss Institue news release describes FRJS:
“The first step of FRJS works like a cotton candy machine — a liquid polymer solution is loaded into a reservoir and pushed out through a tiny opening by centrifugal force as the device spins. As the solution leaves the reservoir, the solvent evaporates, and the polymers solidify to form fibers. Then, a focused airstream controls the orientation of the fiber as they are deposited on a collector. The team found that by angling and rotating the collector, the fibers in the stream would align and twist around the collector as it spun, mimicking the helical structure of heart muscles. The alignment of the fibers can be tuned by changing the angle of the collector.”
It would take more than a century to 3D print every bit of collagen in the human heart at a single micron diameter resolution; it takes a single day with FRJS, according to the Harvard researchers.
The Harvard Office of Technology Development is exploring commercialization opportunities around the intellectual property related to the project.
Said Parker: “This work is a major step forward for organ biofabrication and brings us closer to our ultimate goal of building a human heart for transplant.”





Certainly a large step indeed toward producing a usable replacement heart. But certainly a lot of steps remain.
Verifying the conclusions of half a century ago is interesting as the question is why did it take so long.
So what will the next steps be??
Please keep us informed about this.