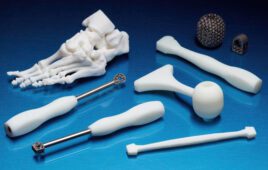
R. Scott Rader is frustrated with the overhyping of 3D printing. Rader, who leads Stratasys’s newly-formed Medical Solutions Group, says his frustration stems from the fact that, as he puts it, “the potential is huge without the hype.”
R. Scott Rader is frustrated with the overhyping of 3D printing. Rader, who leads Stratasys’s newly-formed Medical Solutions Group, says his frustration stems from the fact that, as he puts it, “the potential is huge without the hype.”
As an example, Rader describes a very realistic scenario for orthotics. “I’m not talking about an off-the-shelf device that you can buy today at the drug store. I’m talking about a prescription orthotic that really aims to correct a problem.”
Rader describes the process the way it exists today, which involves a patient seeing a doctor for a prescription. The doctor takes a cast of the patient’s foot and sends the cast through the mail to a manufacturing site. Then the manufacturer sends the device to the office and, after a few weeks, the patient comes come back in for a fitting. This process could take up to a month.
In the practical but infinitely improved world of 3D printing, the doctor sends a foot scan that has been converted into a CAD file to the manufacturer, who then sends the finished device directly to the patient. “There is less need for fitting, because the scan is far more accurate,” says Rader. Further, he notes, that process would take less than a week, and would likely be done within 2 days.
Rader also notes that some people have the aspirational goal to take the system even further, wherein the patient might obtain a prescription and go to a local pharmacy. The pharmacists would access the scan, and then, using the CAD file and a desktop printer, create a device within a matter of hours. That scenario, he says, is a great goal, but “Nothing for that eco-system truly exists today.”
There are risks in such a system that would have to be mitigated, he adds. “There are complex algorithms required to take your foot scan and print a medical orthotic. The quality systems to measure it and make sure that it’s good, as well as somebody to clean it once it comes off the printer – those don’t exist.”
So even without over-promising, 3D printing has potential to revolutionize certain parts of the industry, and many of those opportunities are still in their infancy. As SME’s Lauralyn McDaniel puts it, “Prosthetics and models have been around for 25 years, but some of the scaffolds and implants, are brand new, or are only 3 to 4 years on the market.”
McDaniel organizes the opportunity as follows:
• Prototyping, which is already an established technology.
• Models used in educational, surgical planning, and testing.
• Instruments, including patient-specific cutting guides.
• Bioprinting scaffolds and tissues.
Of those three, McDaniel has personal knowledge of the benefit of patient specific cutting guides. Her friend’s 17-year-old daughter had hip replacement surgery last year. Because of cutting guides created via 3D printing, what is normally a daylong surgery took only 2 hours. She also says that, for implantable orthopedic devices, the manufacturing moves from a multi-step process to a 2-step process because the surface textures can be printed directly onto the implant. OPM, a manufacturer in Connecticut, combines laser sintering additive manufacturing technology with its proprietary OXPEKK material to 3D-print orthopedic and neurological implants.
Measuring matters
There are various technical challenges that industry is working to overcome. And there are limitations to the 3D printing in general. For one, says Rader, when you are working with a 3D printed device, you have to be able to measure the internal cavity. But since each part is printed individually, you cannot use destructive testing or batch testing. For example, consider the tubes that reduce scatter in CT and MRI scanners. These long, thin tubes are created for patient scanning and they are critical to producing a clear image. “So you make these very long, thin tubes, with a challenging aspect ratio – they are a thousand times longer than they are wide.” How do you check the channel, which may be 50 to 100 microns wide?
“It seems like a complex problem,” says Rader. But there is a solution, and Rader says quality assurance simply needs to be aware that the options exist. “You can do acoustic imaging, X-ray based metrology, and CT scanning to visually inspect the internal structures.” Rader says eventually he’d like to see a vision system monitoring part build while it is in the printer. “That’s where we are headed.”
Suppliers such as The Tech Group recommend CT scanner inspection of 3D printed parts, and OEMs might want to consider purchasing a part scanner, depending on their individual needs.
For McDaniel, however, the real problem is not the engineering issues or the metrology challenges, but the need for consistent and better standards. “The biggest challenge right now is the lack of standards and common protocols.”
A question of language
“There are at least five different standards-development organizations working to create these documents,” says McDaniel. “Our big challenge is going to be coordinate as accurately and as quickly as possible, to build evidence for printing in medicine,” she says.
“Right now, at most hospitals, protocols and standards are up to the physician. Right now we have no common language,” she says.
Rader agrees. “Engineers and manufacturers speak a language of machines, materials, tolerances, engineering concepts for stress and strain. Doctors speak a language of, ‘Here is what I want to do and here is the problem that my patient has and I just want to fix it.’” Rader says it takes a tremendous amount of effort, regardless of the manufacturing technology, to bridge the language gap.
Building a case for outcomes
In the grand scheme, Rader notes, only two things count: Economics and outcomes. “We have to make outcomes better and better,” and more importantly, McDaniel points out, we have to be able to show it.
McDaniel also notes that data collection and setting standards for that data are going to be a challenge. “We need to answer the question of what data should be collected,” she says. For example, she says with the surgical cutting guides mentioned earlier, CMS will need to understand the cost and time savings, as well as the improved outcomes. Right now, she says, “the channels of communication are too distributed and there is not enough data for regulations or for reimbursement.”
This type of data collection needs to be specified for all finished devices, but Rader says that’s no different from any manufacturing process, such as injection molding. The process is ongoing.
McDaniel says she is currently expecting the radiology group to already have standards for data “but integrating that into software and printing recommendations is difficult.” McDaniel also mentions that there are 30-40 standards confirmed or in development from various standards development organizations. “It’s not too much, because the technology area is so diverse,” she says. “There are multiple uses, materials, and functions.”
Both SME and Stratasys support collaboration with AmericaMakes, which has developed the Additive Manufacturing Technology Roadmap to identify measurable and meaningful challenges that, when met, promote inquiry, knowledge-sharing, and technical advances across the industry.