As the neurovascular intervention marketplace accelerates toward miniaturization, innovations such as 2.5:1 peelable heat-shrink tubing (PHST), cut-to-length PHST and multi-filar active catheter solutions will come into their own.
Joe Rowan, Junkosha

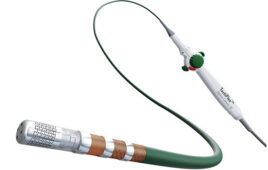
Multi-filar cable (Image from Junkosha)
Catheters are becoming increasingly complex in terms of the procedures they can be used for, making the peelable heat-shrink tubing (PHST) market an extremely exciting area to be involved in. Not only is it helping to address healthcare customers’ unmet needs, but it also paves the way for progressively smaller catheter-based procedures — an ongoing requirement for medical device manufacturers.
The benefit of PHST is that it avoids peeling the outer fluorinated ethylene propylene (FEP) lamination layer, which can damage the catheter within. PHST also enables the final product to be produced more rapidly with improved yields and lower inspection levels while being increasingly ergonomically safe.
Newer innovations are adding to PHST’s capabilities. For example, the unique 2.5:1 PHST solution has been designed to equip catheter manufacturers with the highest shrink ratio currently possible in peelable FEP.
This solution enables them to save time and money through a reduced number of shrink processes. Additionally, thanks to its “take-up,” PHST allows the use of cost-effective, lower-tolerance, baseline materials in the manufacturing process and enables the ability to reflow these easily into a single smooth construct quickly and efficiently. This results in a reduced total cost of ownership for the catheter manufacturer.
There is a wide range of applications for this innovation, including neurovascular catheters that have tapered diameters for the floppy distal segments and proximal sections with larger diameters for pushable support. Because these catheters are mainly being braid-reinforced proximally and coil reinforced distally, they need a PHST solution that can accommodate in a single step the compression required to provide significant bond strength of the materials.
Miniaturization
Aside from 2.5:1 PHST, a greater requirement across the tubing sector is the move toward smaller procedures. Innovations in this area include ultra-small PHST — a tubing suited for laminated jacket coating of tiny guide wires (down to 0.011 in and 0.014 in), leveraging the fact that PHST has a recovered ID down to 0.009 in. These miniature guide wires are perfect for applications including the navigation of vessels to reach a lesion or vessel segment within, for example, the brain or the heart.
Indeed, the trend toward procedures using smaller and smaller catheters is driving the market toward miniaturization. This is accelerating as medtech manufacturers demand catheter solutions that can readily penetrate harder-to-reach places.
The applications that will result from the catheter miniaturization include neurovascular delivery of devices such as coils and stents for stroke or aneurysm therapies, and even signals/energy to help support treatments such as neuromodulation or neurostimulation in the case of research for the treatment of Parkinson’s disease.
Added to this is the requirement for increasingly sophisticated catheters that can send diagnostic signals into the body or provide therapy — in other words, “active” catheters. Such technology is being enabled by innovations including multi-filar, a new fine wire and cable solution designed using precision-engineered PTFE lamination technology. The solution provides multiple signals through an ultra-small shaft, enabling a new generation of small, flexible and intelligent catheters to push the boundaries of current medical procedures.
Single-strand configurations are joined into a multi-filar assembly that can be used in electrophysiology catheters for applying pacing and recording protocols from inside the heart, in ablation and balloon ablation catheters for atrial fibrillation, and in cardiac mapping. The multi-filar technique also allows for easier assembly of the signal or power wires into the final medical device, making the manufacturing process simpler and more cost-effective.
Cut-to-length
Today’s catheter delivery systems rely on increasingly complex shapes, constructions and polymers to deliver their intended treatment. Increasing complexity in catheter bonding and welding applications can only be consistently achieved through advanced technology and proper application of sound engineering principles.
Neurovascular catheters are used to deliver various devices to the brain and feature in numerous cranial procedures. Innovation in the treatment of ischemic stroke with new technology such as stent retrievers and flow-diverting stents will drive an increase in the number and type of catheters required.
This brings with it the need for shorter-length PHST solutions as the length of the catheter that requires the reflow of materials might be shorter. Just like longer forms of PHST, cut-to-length and slit solution enables the catheter manufacturer to use a shortened length to not only reflow the materials, but also to bond, tip and weld complex shapes, sections and segments needed in today’s procedures.
Cut-to-length PHST is also in demand due to the increased availability of custom-engineered diagnostic interventional catheters, which are designed for tougher procedures inside the body. Cut-to-length PHST is perfectly suited for this as it can be cut to a specific size for a given requirement. The cut-to-length and slit option incorporates a peeling slit that allows for faster throughput during manufacture, thus improving cost savings.
Looking ahead
With the push for smaller procedures increasing every year, it is a critical requirement for tubing manufacturers to enable catheters to complete these tasks efficiently and cost-effectively. This challenge can only be overcome through one thing — innovation.
Joe Rowan is the President and CEO for USA & Europe of Junkosha. He brings a wealth of experience in all aspects of developing and profitably growing global high-tech business, with a specialty in advanced fluoropolymers for the medical device industry.
The opinions expressed in this blog post are the author’s only and do not necessarily reflect those of Medical Design and Outsourcing or its employees.