By Sean Madanipour, Medical Design Engineer, Bal Seal Engineering, Inc., Foothill Ranch, Calif.
For some implantable pulse generator makers, integrating connector system components may be the first step on the path toward accelerated development and more breakthrough products.
Active implantables, also known as implantable pulse generators, or “IPGs,” have been widely used since the 1950s. These pulse generators are located within the body and send electrical pulses to nerves and organs. Their earliest application was in the treatment of cardiac-related conditions such as bradycardia (slow heart rhythm) and tachycardia (fast, irregular heart rhythm). But, by 1960, IPGs were delivering pain-reducing, life-improving therapies to the deep brain and spinal cord, as well.
In recent years, advances have been made in the integration, design, and efficiency of IPGs, and many more uses have emerged. Seeking to deliver new modalities of pain management to an aging population, some manufacturers have been introducing new types of IPGs for treating angina, epilepsy, and hearing loss. In some cases, new IPGs can predict seizures by sensing microvolt fluctuations in brain wave activity and potentially stop them before they begin.
A Top-Down Approach
One area identified by many OEMs as ideal for integration is the device header. This implantable-grade plastic or silicone enclosure at the top of the IPG is hermetically sealed to the “can,” or metal housing in which the electronics and battery reside, and it can make up as much as 25% of the overall device volume. All of the critical connections that drive the IPG’s functions originate in the header connector cavity where power is transmitted through multi-channel connections as signals from the battery and electronics to the lead, and ultimately out to the body. Electrical contacts inside the header ensure that the proper signals get transmitted through the right channels on the lead, and seals between those contacts isolate against both bodily fluids and electrical “cross-talk” that may otherwise cause false positives, improper stimulation, or general malfunctioning of the device.

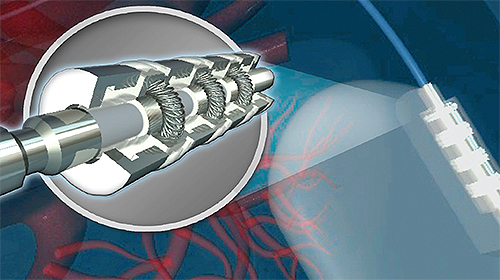
The SYGNUS™ Implantable Contact System for IS-4 and DF-4 standards consists of three contacts and four seals, as well as distal and proximal caps.
In the half-century or so since the introduction of the first IPG, the dual need for connecting and sealing has remained constant. But some of the tools engineers use to meet that need have changed significantly.
The Connection Progression
In the early days of IPGs, a setscrew was the primary method for fastening and connecting. Used in series along the header, it provided both mechanical retention and electrical contact. But as device functionality improved and the need for multiple connections grew from four to six, or as many as 16 in some neurostimulators, the process of tightening each setscrew during the surgical process became less and less viable. While it acknowledged the continued need for a setscrew to ensure absolute retention of the lead in the device header, the medical community demanded alternatives, and several new types of contact approaches began to emerge.
Among the first of these to supplant the multiple setscrew setup was the Multi-Beam contact. Resembling a leaf-spring ribbon inside a can, this contact was designed, manufactured and introduced by Medtronic, and it is still widely used today. Constructed of stainless steel, Titanium and MP35N, the Multi-Beam offers cost-effective electrical connectivity. However, its relatively low number of contact points (6 to 8) can mean higher contact resistance, which may reduce battery and device service life. In addition, the Multi-Beam has inherent size limitations, and its leaf springs have linear force curves, which can produce variable insertion and extraction forces.
The Canted-coil ™ spring, a contact developed around the same time as the Multi-Beam, offered designers yet another new alternative to the setscrew. Designed and patented by Bal Seal Engineering, Inc., the spring was first employed in an IS-1 ring connection, but it soon evolved into a spring and housing combination that could provide a desirable weld surface. The combination, which became known as the Bal Conn™, continues to be specified by many of today’s device makers, due in large part to its ability to provide very low electrical resistance in a limited space. The Bal Conn’s Platinum-Iridium spring allows for low, highly-controllable insertion and extraction forces, and its spring coils provide multi-point conductivity. While it can help conserve header real estate, the Bal Conn is best suited for challenges that can’t be met by off-the-shelf types.

By using standard, integrated seal and contact assemblies, some startups and mid-level device manufacturers have been able to accelerate development and reduce device volume.
The ImplanTac® Contact Socket is a more recent IPG interconnect. Its manufacturer, Hypertronics, describes it as a “basket of wires” design that positions lengths of wire strung at an angle to the socket’s axis. This contact offers low electrical resistance and low insertion and extraction forces, but its construction limits it to a set axial length, which can be prohibitive for OEMs seeking to reduce package size or pack more contacts into the connector cavity.
Stuck in the Middle
Unlike their contact counterparts, the high dielectric-strength seals used to provide fluidic and electrical isolation inside the device header haven’t evolved all that much. Generally, they have been and continue to be molded from implantable-grade silicone, a material with properties that are widely known and accepted in the implantable world. Silicone is soft and malleable, so it flows easily into and around non-uniform surfaces while retaining the ability to seal. It is also relatively inert, so it’s less likely to react with the metals used to form the contacts and lead.
Arriving at Integration
Traditionally, IPG producers have had little choice but to undergo the costly, iterative and time-consuming job of purchasing electrical contacts and designing, creating, and testing their own isolation seals for the critical lead interface. By some industry accounts, this process can tack as much as six months onto the development of a new device.
However, a new kind of “plug-and-play” connector introduced in October 2010 may make the process obsolete – at least for some smaller OEMs and startups focused on emerging therapies. The concept behind this new technology is relatively simple: Take a proven electrical contact platform and package it with isolation seals that can guard against fluid infiltration and prevent signal leakage. Then, test the components for insertion and extraction forces, conductivity, electrical resistance, seal isolation and other criteria, so that a manufacturer can confidently integrate the entire connector system into its device design and move closer to the ultimate goal of market introduction.
An example of this new approach, which effectively pairs seals and contacts in a pre-tested, compact and scalable package, has been developed by Bal Seal Engineering, maker of the original Bal Conn. Billed as the “world’s first seal-integrated contact system” and marketed under the tradename SYGNUS™, Bal Seal’s new solution is built around its Canted-coil™ spring system.

In this cutaway of an integrated seal/contact assembly designed for compliance with ISO’s IS-4 standard SYGNUS™ for IS-4, the electrical contact housings interlock with silicone isolation seals.
The SYGNUS system combines the tiny springs made from Platinum-Iridium, metal housings of MP35N® and implantable-grade silicone isolation seals in a configuration that forms a dense connector “stack” to accommodate lead diameters ranging from 0.90mm to 3.20mm. Since a version of the seal and contact combination is engineered to the recently-ratified four-pole standard (IS-4 and DF-4) for cardiac health management devices (a proximal cap serves as the datum, and a distal cap completes the footprint), the company says SYGNUS gives manufacturers the ability to dedicate resources to therapy and function improvements, instead of component procurement or development.

An integrated connector system approach makes it easier to scale the number of seals and contacts up or down, based on application requirements.
Stack configurations like the one pictured above can be found in many neurostimulation devices.
Cardiac IPG device makers who have recently tested and adopted the new standardized seal and contact technology cite reliability, safety and ease of implementation as primary reasons for their choice. And in a move that mirrors their CHM counterparts, designers of active implantables used in the delivery of neuromodulation therapies are beginning to embrace the same kind of stack solution, engineered to even smaller, more contact-dense specifications, for the very same reasons.
In 2009, CCC Medical Devices, a prominent neuromodulation and cardiac IPG design and contract manufacturer based in Uruguay, began evaluating an integrated connector system approach. In early 2010, the company re-examined its old method of dual sourcing for some of its products, and replaced individual seal and contact components with Bal Seal’s SYGNUS system. Based on its success, CCC is considering the same approach with more IPG designs in the future.
Oscar de Oliveira, an electrical engineer and CCC’s Manager of Business Development, says the switch positively impacted both technology and yield.
“Choosing an integrated seal and contact system enabled us to significantly improve the assembly process,” says Oliveira. The all-in-one SYGNUS design we use not only ensures electrical insulation between each contact, it also helps eliminate epoxy leakage that could occur during header casting. For us, that’s less costly scrap and better, more productive manufacturing.”

A SYGNUS integrated connector system is clearly visible in the header of this IPG, used to deliver neurostimulation therapy.
Oliveira adds that an important side benefit of the SYGNUS seal/contact system he’s using is its ability to reduce header size.
“The header volume is usually 1/4 to 1/5 of the total IPG volume, so any solution that helps to reduce the header will have an important impact on the IPG size,” Oliveira says. “The system’s compact footprint has helped shrink header volume while keeping a good insulation between contacts.”
For now at least, this new focus on systemization – especially of the contact/seal variety – is more prevalent among small, agile manufacturers whose organizations can’t afford the vertical integration, which is common to the industry giants. But it may not be long before even the heavy-hitters discover the benefits of specifying pre-packaged, pre-tested connecting solutions for their devices.
“There’s definitely a growing need for this kind of solution,” says Bal Seal Business Development Manager, Bill Nissim. “To some companies, especially those facing venture capital deadlines and clinical trial windows, choosing an integrated system provides a level of freedom that could mean the difference between success and failure. Similarly, from a patient’s perspective, it might be one of the factors that determine whether or not a breakthrough therapy ever makes it to market.”
Discuss this on The Engineering Exchange:
CCC Medical Devices
www.ccc.com.uy/
SYGNUS
www.sygnusconnects.com
::Design World::




