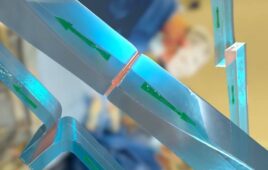
Medical device-maker Instratek chose Solvay Specialty Polymers’ glass-filled Ixef® 1022 polyarylamide (PARA) resin to mold several components of its single-use STAPiX™ staple fixation system for surgical applications targeting the forefoot, mid-foot, hind-foot and hand. Photo courtesy Instratek.
Solvay Specialty Polymers, a leading global supplier of high-performance thermoplastics, recently announced that Instratek, a Houston-based developer of medical devices for extremity surgery, selected Solvay’s Ixef 1022 polyarylamide (PARA) resin to mold several components of its STAPiX staple fixation system used for surgical procedures on the forefoot, mid-foot, hind-foot and hand. Instratek, which just launched the STAPiX system in the United States, chose Ixef PARA resin for its high strength and stiffness, excellent moldability and attractive price point for single-use medical devices.
Ixef 1022 PARA, a 50 percent glass-filled compounded resin, is used for the staple inserter handles, which are subject to high bending stress when the device opens a superelastic nitinol staple prior to its insertion into the bone. Of the polymers tested, Ixef 1022 PARA best minimized flexing in the tool’s handles while bending the nitinol staple.
As with all of Solvay’s medical-grade polymers, Ixef 1022 PARA demonstrates no evidence of cytotoxicity, sensitization, intracutaneous reactivity or acute systemic toxicity, based on biocompatibility testing as defined by ISO 10993:1. Medical-grade Ixef PARA resins are available worldwide in a broad range of gamma-stabilized colors, making them well-suited for single-use medical device applications that must maintain excellent aesthetics despite repeat sterilization by gamma radiation or other methods, such as application of ethylene oxide.
Solvay Specialty Polymers has been a reliable materials supplier to the healthcare industry for more than 25 years. The company is a leading manufacturer of healthcare polymers, offering the industry’s broadest range of high-performance polymers for medical devices, instruments, and equipment. Solvay also offers a family of Solviva Biomaterials for use in a range of implantable devices.
Solvay Specialty Polymers
www.solvayspecialtypolymers.com