Compliance to regulatory standards is an absolute priority for manufacturers of medical devices. Non-compliance is simply not an option for this market area.
This article explains how modern Manufacturing Execution Systems (MES) help with this compliance and keep operations running smoothly and efficiently.
A MES will facilitate configuration, tracking, history, and compliance of software throughout a factory. As part of its many capabilities, it will track manufacturing processes, components, equipment, and people in order to enable effi cient resource management and production scheduling. It will control and document production processes to show that these have been carried out correctly to comply with industry standards and government regulations. Compliance involves identification of the product being made, including what components are being used, and who the people are who are working on it. It requires verification steps to ensure the correct processes are being utilized. Any exceptions from the norm need to be documented and appropriate action taken. Everything that is happening needs to be clearly documented and time stamped.
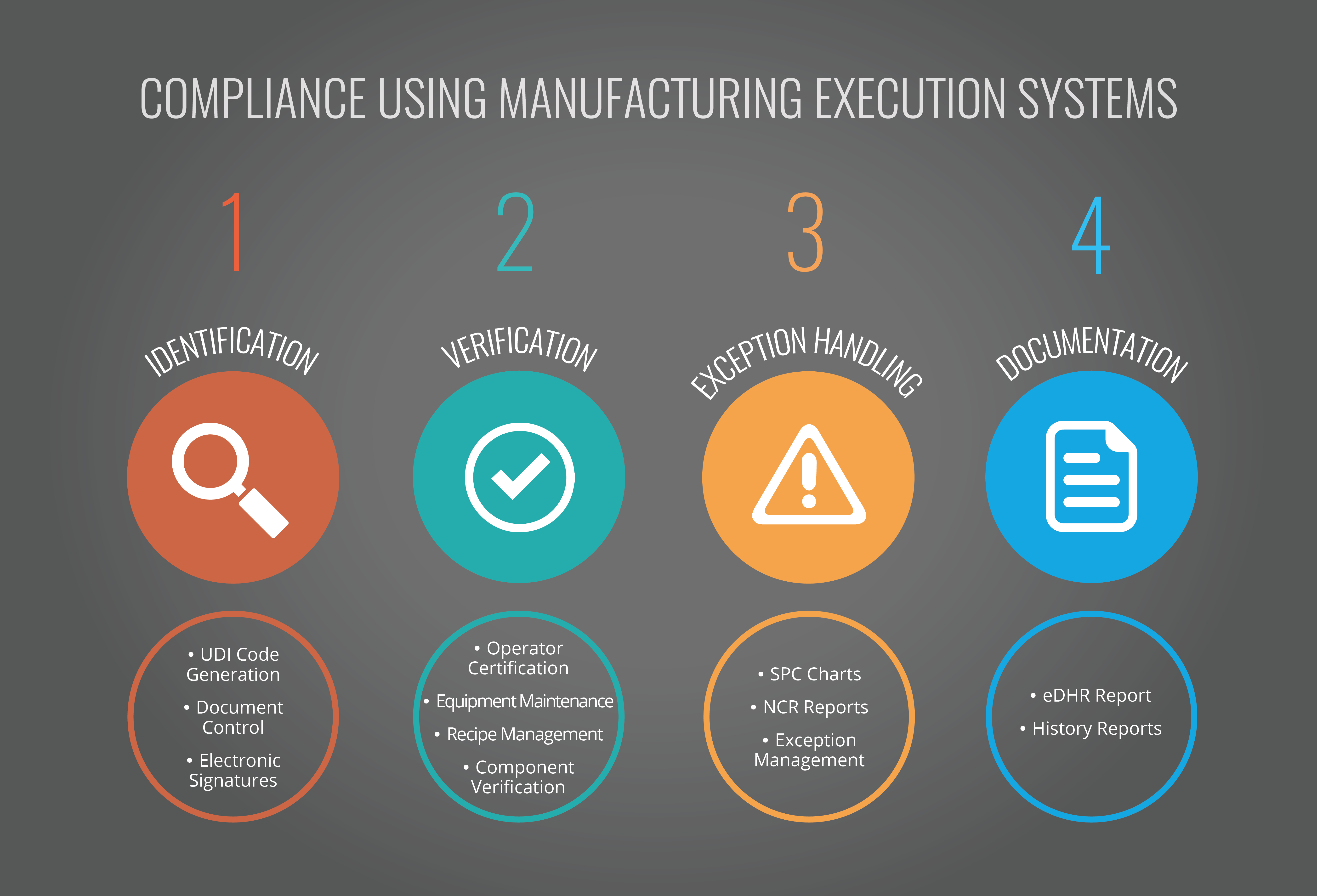
Depending on the local regulations and devices being produced, records may need to be stored for anywhere from 20 to 50 years. To help achieve compliance, MES can help in all four of these areas: Identification, verification, exception handling, and documentation.

(Figure 1. Compliance and Traceability Made Easy with Manufacturing Execution System)
Identification
Unique device identifier (UDI) codes are required by The U.S. Food and Drug Administration (FDA) to track unit assembly. A MES can automatically generate a UDI that can be utilized in processing, associated with the history of a particular product batch, and used to identify the unit through any post-manufacturing processing. Documents pertaining to procedures, processes, equipment, regulations etc., can also be version controlled and stored within the MES. They can be attached at any point of the processing stream. The document can be just a reference or a requirement that the operator must review prior to executing a particular process.
At any stage during manufacture, the MES can further request that the operator “sign” that they have performed a task. This form of electronic signature includes the name of the operator and a password only known to them to uniquely identify them as having carried out the task. The system handles control of passwords in line with 21 CFR Part 11 including password ageing, automatic log-off, and password quality.
Verification
The MES can offer complete control over the verification that all processes, personnel, and machinery are as they should be. It can incorporate and record operator certifi cation, equipment maintenance records, recipe management, and verification that correct components are being used.
At any stage in a process, an MES can request verification that the specifi cations of parts or components are within limits. This can include capturing values of process variables that will be kept as part of the batch record. Values can be checked against tolerance limits and if they fall outside of specification limits appropriate corrective action can be taken in line with the exception handling procedure
(Figure 2. Achieving Compliance Using Manufacturing Execution Systems)
Exception Handling
Statistical Process Control (SPC) is an integral part of a MES. If a part moves outside of its specifi cation limits, the MES helps the operator evaluate the situation by generating an SPC chart which contains the data associated with the anomaly. The Critical Manufacturing MES displays SPC charts which document the ranges, chart point details and statistics pertinent to the anomaly. This includes chart statistics which show how often specification limits are being exceeded. It also generates a built-in out-of-control action plan process that can automatically put a product or batch on hold.
In this case, an exception management processing plan must be performed before the product can progress any further through the manufacturing process. The MES can also be used to create non-conformance reports (NCR) based on a template configured to meet an organisation’s requirements. This can record any required information about the material exception including severity, production order, and associated error codes. Annotation of the electronic device history report (eDHR) can include the NCR number, date, time, and can provide an electronic signature for the originator of the NCR event. The exception management process within the MES can be completely customized to define a methodology to meet a factory’s specific business requirements. This gives absolute control over the NCR and exception management process with complete audit trail and visibility. Documentation
The eDHR is the most critical document that the FDA requires and provides necessary evidence that manufacturing processes have been correctly carried out. The MES automatically generates this document along with other history reports, recording all steps and data as an order moves through the factory to completion.
MES Adds Flexibility & Efficiency
Through complete visibility, tight process control, reduced production variability, traceability, and secure recording of all information relating to the production process, the MES provides solid support for compliance to FDA titles 21 CFR Part 11 and 820. In addition, it can also help improve production efficiency and reliability; increase manufacturing yield, and lower costs. Modern MES can provide manufacturers with a solution that offers high availability and total flexibility to handle even the most sophisticated processes, production flows and models while still ensuring complete traceability of batches even when they are split or merged. It can also adapt to changing processes, ensuring that effi ciency is maximized and innovation not compromised. Automating data collection, calculations, and parameter deviation control delivers signifi cant increases in production efficiency and reduction of Figure 2. Achieving Compliance Using Manufacturing Execution Systems Emphasis On Contract Manufaturing human error. The volume of data collected also enables greater analysis of processes to further drive efficiency and deliver predictable, high quality results. MES makes this possible thanks to its powerful integrated analytical tools which can be seamlessly connected to business systems for complete visibility.
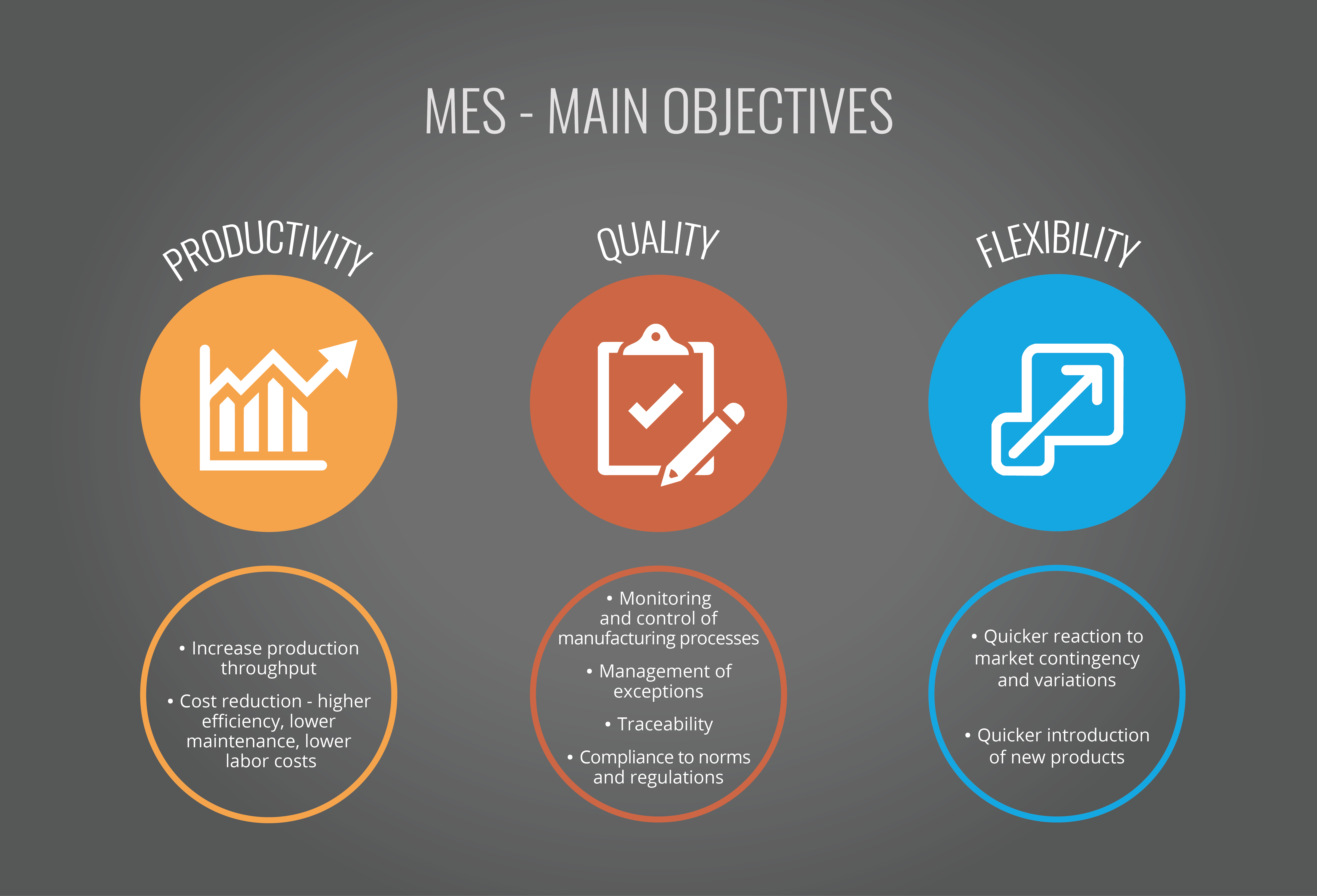
(Figure 3. MES – main objectives)
MES for the future
The world of manufacturing is changing as it responds to demands for smaller, higher mix batches and customized solutions. MES technology, provided by vendors like Critical Manufacturing, can deliver these benefi ts to Medical Device manufacturers while giving all the support needed for compliant operations. These systems integrate computational networks with the surrounding physical world and its processes. They embrace technologies such as embedded computing in components, mobile computing technologies, the Cloud and the Industrial Internet of Things (IIoT).
Through the use of de-centralized models for control of manufacturing execution, they enable production of custom products with high levels of efficiency. For Medical Devices, this opens up greater possibilities for patient-specific devices. Using embedded computer technology, both components and equipment can have their own intelligence and the ability to make autonomous decisions. This can even include deciding for themselves the most efficient route to take through manufacturing. While this offers many opportunities for dynamic automation and ultimate production effi ciency, care also needs to be taken that the model still retains the required functionality for compliance with regulations. The IIoT enables all data to be stored for the eDHR and makes this report readily available for review by customers or auditors.
Adding communication capability to individual products or components also offers the possibility to extend traceability within the MES to the wider supply chain. Such end-to-end track and trace capability results in a highly integrated supply chain that is transparent and more easily auditable while being more reliable and agile.
Summary
For medical device manufacturers, modern MES systems offer an excellent support tool for compliance while providing an excellent return on investment through improved production effi ciency, yield, and reliability. They provide a platform for the future that will help businesses remain competitive to changing market demands and enable the economical production of custom devices. They further give a new level of production flexibility and agility while keeping control of processes and effi ciently handling data for assured quality and regulatory compliance.


