Developing the product requirements is one of the most complex activities associated with product development, yet one of the most important.
Bill Betten, Betten Systems Solutions

[Image, from Flickr, is marked as public domain]
As a reminder, this series focuses on the definition and execution of product development activities post-funding and includes the following:
- Idea – Without it, nothing to be developed
- Process – The structure for development
- Plan – The blueprint
- Requirements – The details (The article you’re reading.)
- Regulatory/ reimbursement – Critical to the medical device space
- Verification/validation – The right product doing the right thing
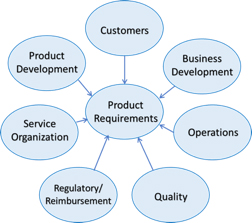
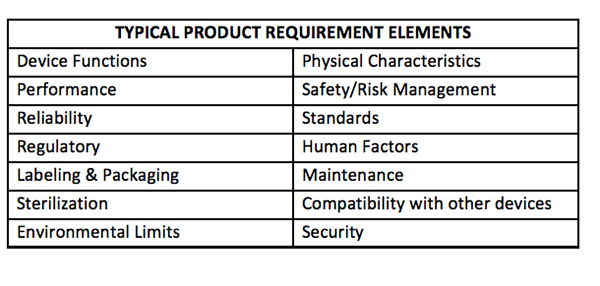
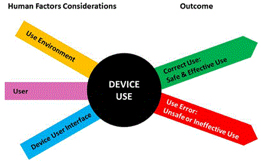
The requirements will form the basis not only for the product design, its features, functions, and risk mitigation, but extend completely through to the verification and validation process, subject of a future article in this installment. To that end, requirements should be clear, traceable, and verifiable. Other things to keep in mind during the development of requirements include the following:
- Involve all the stakeholders in the process of defining the problem and the requirements
- Complete the problem statement before defining the requirements
- Avoid stating the problem in terms of solutions
- Identify the high-level system functions
- State the requirements clearly and unambiguously
- Identify mandatory versus tradeoff requirements and quantify and prioritize if appropriate
Medical manufacturers do not have the luxury of launching products to gather user feedback after launch or to make changes quickly, unlike tactics commonly used in the consumer space, particularly in software-based products. The medical regulatory environment requires tracing from initial requirements to demonstration that those requirements have been met, certainly a demonstration of rigor not mandated in industrial and consumer productions. In addition, the post-launch monitoring requirements that exist regarding medical devices ensures that product issues and deviations from desired performance are identified, tracked, and resolved.
Development of solid requirements should be the confluence of inputs from the business, user and technology constituencies. Even in a development environment that stresses rapid iteration, i.e. “fail fast,” the development and refinement of requirements will enable you to “succeed quickly,” reducing expensive and time-consuming iterations late in the product development cycle. For consumer products, incorrect requirements may lead to expensive changes after products have launched. For regulated medical devices, incomplete or inaccurate requirements may lead to delays in approval, potential recalls, or worst case, harm to the patient. Time taken to define the requirements well at the beginning will ensure that your journey will be swifter and more efficient.
Next up, we’ll examine how the two “Rs” – regulatory and reimbursement – influence the development of your product and need to be an integral and early part of your product development process>>
Bill Betten is the president of Betten Systems Solutions, a product development realization consulting organization. Betten utilizes his years of experience in the medical industry to advance device product developments into the medical and life sciences industries. Betten most recently served as director of business solutions for Devicix/Nortech Systems, a contract design and manufacturing firm. He also served as VP of business solutions at Logic PD, medical technology director at TechInsights, VP of engineering at Nonin Medical, and in a variety of technology and product development roles at various high-tech firms.
The opinions expressed in this blog post are the author’s only and do not necessarily reflect those of MedicalDesignandOutsourcing.com or its employees.