A method to rotate single particles, cells or organisms using acoustic waves in a microfluidic device will allow researchers to take three dimensional images with only a cell phone.
Acoustic waves can move and position biological specimens along the x, y and z axes, but for the first time researchers at Penn State have used them to gently and safely rotate samples, a crucial capability in single-cell analysis, drug discovery and organism studies.
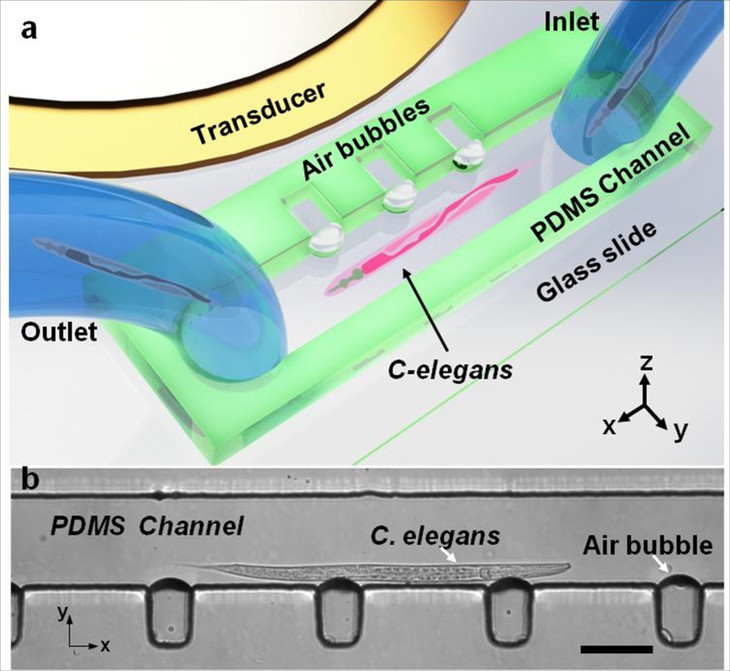
Design and operation of the acoustofluidic rotational manipulation (ARM) device. (a) A schematic of the experimental setup. The piezoelectric transducer that generates acoustic waves is placed adjacent to the microfluidic channel. The acoustic waves actuate air microbubbles trapped within sidewall microcavities. (b) An optical image showing a mid-L4 stage C. elegans trapped by multiple oscillating microbubbles. Scale bar = 100 μm. (Credit: Tony Jun Huang/Penn State)
The research, published today in Nature Communications, was led by Tony Jun Huang, professor of engineering science and mechanics and Huck Distinguished Chair in Bioengineering Science. Huang and his group created an acoustofluidic rotational manipulation (ARM) method that traps bubbles in a series of small cavities inside a microfluidic device. Acoustic transducers similar to ultrasound imaging transducers create an acoustic wave in the fluid, making the bubbles vibrate, which creates microvortexes in the flowing liquid that are tunable so the sample rotates in any direction and at any desired speed.
“Currently confocal microscopes are required in many biological, biochemical and biomedical studies, but many labs do not have access to a confocal microscope, which costs more than $200,000,” said Huang. “Our ARM method is a very inexpensive platform and it is compatible with all the optical characterization tools. You can literally use a cell phone to do three-dimensional imaging.”
To demonstrate the device’s capabilities, the researchers rotated C. elegans, a model organism about a millimeter in length frequently used in biological studies. They also acoustically rotated and imaged a HeLa cancer cell.
Existing methods of manipulating small objects depend on the optical, magnetic or electrical properties of the specimen, and/or damage the specimen due to laser heating. The ARM method, on the other hand, uses a gentle acoustic wave generated by a power similar to ultrasound imaging, and at a lower frequency. The device is also compact and simple to use.
“Our method is a valuable platform for imaging and studying the effect of rotation at the single cell level,” said co-lead author Adem Ozceki, graduate student in engineering science and mechanics. “More important, with the capacity to rotate large numbers of cells in parallel, researchers will be able to perform high-throughput single-cell studies. “
In addition to its applicability to a large range of biological and physical science investigations, ARM technology shows excellent biocompatibility in a HeLa cell viability test in which 99.2 percent of cells survived manipulation.




