 Scientists in Cambridge, Mass. and Chennai, India, are touting self-organizing brain tissue growth in a 3D-printed system.
Scientists in Cambridge, Mass. and Chennai, India, are touting self-organizing brain tissue growth in a 3D-printed system.
Published results in Biomicrofluidics highlight the work of the Massachusetts Institute of Technology (MIT) and Indian Institute of Technology Madras scientists, who have grown small amounts of the self-organizing brain tissue, known as organoids, in a tiny 3D-printed system that allows for observation while they grow and develop, according to a news release.
Technology for real-time observation of growing organoids currently involves commercial culture dishes with many wells in a glass-bottomed plate placed under a microscope. The plates are costly and only compatible with specific microscopes while they don’t allow for the flow or replenishment of a nutrient medium to the growing tissue, the researchers say.
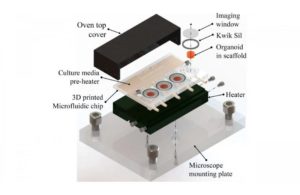
Advances have led to microfluidics, which delivers a nutrient medium through small tubes connected to a tiny platform or chip. Still, there are manufacturing challenges and high costs associated with these. The researchers say their method uses 3D printing to create a reusable and easily adjustable platform that costs just $5 per unit to produce.
The system uses a biocompatible type of resin used in dental surgery, while the printed chip was cured by exposing it to UV light before sterilizing it and placing live cells in the wells. After the wells are sealed at the top with a glass slide, the nutrient medium and drugs used in the study are added through small inlet ports.
To test the device, the researchers used organoids derived from human cells and observed them with a microscope, successfully following their growth and development for seven days. Additionally, the percentage of cells in the organoid’s core that died during that period was smaller in the 3D-printed device than in regular culture conditions, while the researchers believe their cell design protects the tiny growing brain.
“Our design costs are significantly lower than traditional petri dish- or spin-bioreactor-based organoid culture products,” study author Ikram Khan said in the release. “In addition, the chip can be washed with distilled water, dried, and autoclaved and is, therefore, reusable.
“One advantage offered by our microfluidic device is that it allows constant perfusion of the culture chamber, which more closely mimics a physiological tissue perfusion than conventional culture, and thus reduces cell death at the organoid core.”




