[Image from Medical Microinstruments]
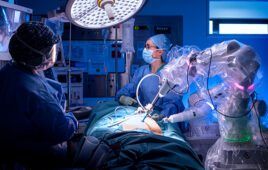
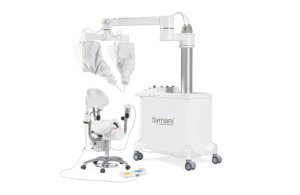
Italy-based MMI designed the Symani system as a flexible platform with two robotic arms that can be easily positioned for surgical procedures across any anatomical region.
Symani has a 7-20X motion scaling with tremor filtration for microsurgery and super microsurgery. Surgeons can scale hand movements while articulating the NanoWrist robotic microinstruments. NanoWrist has seven degrees of freedom for optimal dexterity inside of minimally invasive incisions.
The reconstruction procedure was performed at Careggi University Hospital Florence by Dr. Marco Innocenti and was the first enrolled patient in the Italy-based company’s post-market study. The young adult in the procedure was a candidate for a free-flap reconstruction to save the arm after amputation was recommended. The robotic-assisted procedure successfully restored blood flow to the area despite severe damage and extensive scar tissue inside the arm, according to the company.
“Robotic-assisted technology is a revolutionary tool for microsurgery, because it allows physicians to complete procedures that are difficult or impossible with the human hand,” Innocenti said. “By increasing our precision, we can reconnect submillimeter vessels and nerves that enable reconstruction even after severe trauma. I am confident our post-market study will show significant advantages to improving patient outcomes, as the patient in the motorcycle accident would have lost her arm without robotic-assisted microsurgery.”
The Symani Surgical System won CE mark approval in 2019. It’s meant to enhance free-flap reconstructions, replantation, congenital malformations, peripheral nerve repairs and lymphatic surgeries. It reduces the occurrence of thrombosis by 50% in preclinical studies, according to MMI.
“By evolving surgical intervention from a manual process to one that leverages robotic technology, we can open the door to procedures that have never been done before,” CEO Mark Toland said. “Prof. Innocenti’s accomplishment, in this case, is an extremely exciting piece of validation for our technology that will contribute to our data collection for our growing global regulatory strategy.”