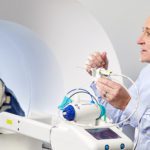
Monteris Medical (Plymouth, Minn.) has brought in nearly $11 million from its latest investment round as it expands sales and marketing of its NeuroBlate robot-assisted brain surgery device, according to a new Form D filed with the U.S. Securities and Exchange Commission.
Monteris Medical (Plymouth, Minn.) has brought in nearly $11 million from its latest investment round as it expands sales and marketing of its NeuroBlate robot-assisted brain surgery device, according to a new Form D filed with the U.S. Securities and Exchange Commission.
Monteris had sought roughly $17 million in total from the money-raise.
The company has overcome challenges after starting 2018 with FDA labeling a recall as Class I. The recall involving unexpected heating and damage to the tip of the NeuroBlate system’s probe when it interacted with an MRI. Monteris Medical resolved the heating issue in October when it received FDA clearance for a laser probe with fiber optic controlled cooling to use in the NeuroBlate system. The probe replaced a metal thermocouple inside the laser probe, and enabled Monteris to lift MR scan restrictions. All patient-contacting components are now non-metallic.
Since resolving problems with the system, Monteris has had a string of good news to report. More than 2,000 patients have been treated with NeuroBlate since its release in 2013, and the company near the end of 2018 also won reimbursement from Aetna and Anthem. CEO Marty Emerson told Medical Design & Outsourcing last month that he is optimistic that the roughly $10 million a year company – which had seen annual revenue growth of 40% before 2018 – is set to grow.
NeuroBlate uses a robot-guided laser to ablate brain tissue during MRI scans. Some brain surgeons find NeuroBlate a useful surgical option for certain epilepsy and brain cancer patients who don’t have many other alternatives, according to Emerson.