SPR Therapeutics received clearance from the United States Food and Drug Administration (FDA) to commercialize the SPRINT Peripheral Nerve Stimulation (PNS) System.
SPRINT is a completely reversible and minimally-invasive peripheral nerve stimulation system to provide relief of chronic and acute pain, including post-operative and post-traumatic pain.
“The FDA’s clearance comes at a critical time when physicians, patients and the U.S. healthcare system are seeking therapies to manage pain while reducing opioid use,” said Maria Bennett, SPR Therapeutics Founder, President and CEO. “SPR has developed a drug-free treatment for relieving chronic and acute pain.”
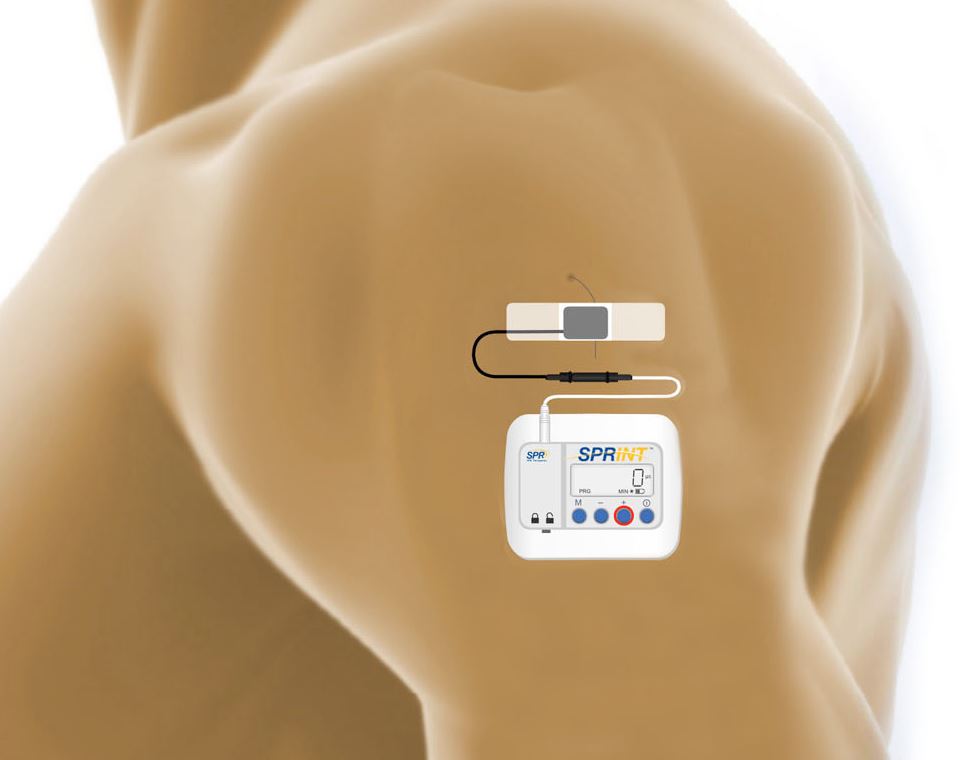
Image: SPR Theraputics
The SPRINT PNS System includes a threadlike, coiled wire, or lead, and a matchbox-sized wearable stimulator, about the weight of four quarters. The lead is placed percutaneously, or through the skin, via a fine needle and connects externally to the wearable stimulator. The stimulator delivers electrical stimulation through the lead, which activates peripheral nerves to achieve pain relief.
SPRINT allows lead placement as far as two to three centimeters from the targeted nerve. This simplifies the lead placement procedure for physicians and gives them the distinct ability to preferentially stimulate specific fibers of the nerve to maximize pain relief. This approach to PNS is fully reversible, as the device is designed to be withdrawn without surgery at the end of the 30-day treatment period.
A physician can place the system during a short outpatient procedure without surgery or anesthesia. Because SPRINT is minimally invasive and completely reversible, physicians may use it earlier in a patient’s treatment process in an effort to reduce or eliminate the need for opiates and to avoid costly alternatives in managing acute and chronic pain.
SPRINT will soon be available at select health care facilities in the United States.