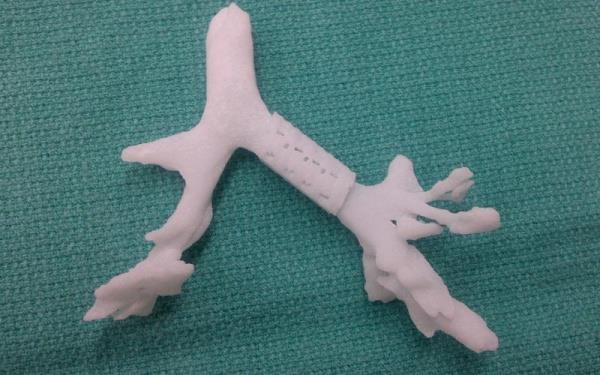
A 3D printed tracheal splint
Today, Materialise and Tissue Regeneration Systems (TRS) announce a partnership to manufacture life-saving 3D-Printed tracheal splints for use in clinical trials.
Materialise’s Mimics Innovation Suite was used to design the splint which is constructed from a bioresorbable technology platform licensed to TRS by the University of Michigan in 2007. After several years refining fabrication methods, TRS received its first commercial product clearance from the FDA in 2013. The Materialise partnership is one of several co-development projects currently underway at TRS.
Thanks to FDA approval for Expanded Access to an investigational medical device, the splints are used to treat tracheobronchomalacia (TBM), a condition affecting 1 in 2,220 babies which can cause the airway to spontaneously collapse.
“The collaboration between TRS and Materialise will provide production capacity for the tracheal splint, which will allow the splint to be available to a larger number of infants who are affected with TBM,” stated Bryan Crutchfield, Managing Director, Materialise USA.
“We are excited to have the opportunity to impact the lives of children affected with TBM through the production of the tracheal splint,” says Jim Fitzsimmons, CEO of TRS.




