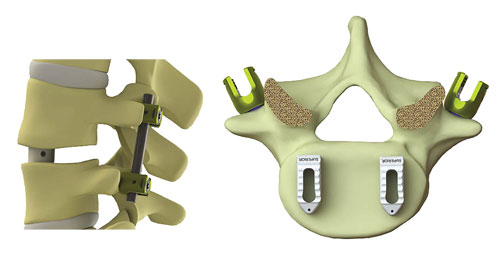
The medical industry continues to grow in importance for design engineers. Helping to spur growth is the introduction of a number of materials for devices and applications. One of those materials is polyetheretherketone (PEEK) and its variations. Nexxt Spine LLC, a Fishers, Ind.-based manufacturer of spinal implants and instrumentation, recently received FDA clearance for its Honour™ cervical spacer and lumbar spacer implants made of Zeniva® polyetheretherketone (PEEK) rods from Solvay Specialty Polymers USA, LLC. Zeniva® PEEK has a modulus very close to that of bone plus excellent toughness and fatigue resistance.
The Honour™ Spacer System consists of three unique implants offered in various footprints, with indications ranging from the cervical to the lumbar spine. The system can accommodate unilateral (TLIF and oblique) and bilateral (PLIF) approaches in the lumbar spine with the option to insert directly or initially on its side to prevent over-distraction of the ligamentous and nerve structure. The spacer systems are hollow so that bone can grow through, fusing the adjacent bony surfaces of the vertebrae.
Zeniva® PEEK offers advantages over metals such as titanium for intervertebral implantable devices. These advantages include biocompatibility, chemical inertness, and a modulus of elasticity that is close to that of bone. Based on biocompatibility testing, Zeniva® PEEK demonstrates no evidence of cytotoxicity, sensitization, irritation, or acute systemic toxicity. It also has high strength and stiffness and radiolucent properties that permit x-ray procedures.
Zeniva® PEEK and the entire line of thermoplastic Solviva® Biomaterials are manufactured in compliance with the relevant aspects of ISO 13485 and under the relevant aspects of current Good Manufacturing Practices. In addition, all materials are tested in an accredited lab that is ISO 17025 compliant.
Solvay Specialty Polymers
www.solvayspecialtypolymers.com