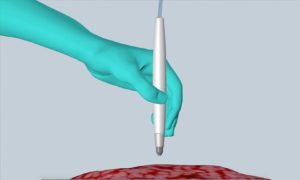
The MasSpec Pen rapidly and accurately detects cancer in humans during surgery, helping improve treatment and reduce the chances of cancer recurrence. [Image from University of Texas at Austin]
A team at the University of Texas at Austin has invented a tool used during surgeries that can give results in about 10 seconds, which is 150 times faster than current technology, according to the researchers. The device, known as the MasSpec Pen, is a handheld instrument that can give surgeons precise diagnostic information about which tissues should be cut or saved to help improve treatment and reduce the likeliness of cancer returning.
“If you talk to cancer patients after surgery, one of the first things many will say is ‘I hope the surgeon got all the cancer out,’” Livia Schiavinato Eberlin, assistant professor of chemistry at UT Austin and leader of the research team, said in a press release. “It’s just heartbreaking when that’s not the case. But our technology could vastly improve the odds that surgeons really do remove every last trace of cancer during surgery.”
Currently, the way for diagnosing cancers and figuring out the boundary between cancer and normal tissues in surgery is the Frozen Section Analysis. That method tends to be slower and less accurate than the UT Austin method. Frozen Section Analysis results can take 30 minutes or more to prepare results that a pathologist can interpret, which could also increase the risk of infection and negative effects of anesthesia. Additionally, Frozen Action Analysis can be inaccurate for certain types of cancer in as many as 10 to 20% of cases.
The MasSpec Pen was tested on tissues removed from 253 human cancer patients. It took about 10 seconds for a diagnosis to occur and was shown to be more than 96% accurate. The device could also detect cancer is marginal regions in normal and cancerous tissues the had a mixed cellular composition.
“Any time we can offer the patient more precise surgery, a quicker surgery or a safer surgery, that’s something we want to do,” James Suliburk, head of endocrine surgery at Baylor College of Medicine and a researcher on the device, said. “This technology does all three. It allows us to be much more precise in what tissue we remove and what we leave behind.”
The device works by seeking out metabolites. Metabolites are small molecules that living cells produce, regardless of if they’re healthy or cancerous. Each cancer type makes a unique set of metabolites and other biomarkers that tend to be like fingerprints.
“Cancer cells have dysregulated metabolism as they’re growing out of control,” Eberlin said. “Because the metabolites in cancer and normal cells are so different, we extract and analyze them with the MasSpec Pen to obtain a molecular fingerprint of the tissue. What is incredible is that through this simple and gentle chemical process, the MasSpec Pen rapidly provides diagnostic molecular information without causing tissue damage.”
Molecular fingerprints picked up by the MasSpec Pen are evaluated by statistical classier software that is trained on a database of similar fingerprints that Eberlin collected from 253 normal and cancerous tissue samples. When the MasSpec Pen finished its analysis, “Normal” or “Cancer” appear on a computer screen automatically. Other types of cancer could also show a subtype.
The device is handheld, making it easy for physicians and surgeons to operate. All they have to do is hold the pen against a patient’s tissue which will trigger an automated analysis using a foot pedal. After 10 seconds, the results will appear. During the process, the pen puts a drop of water on a tissue that attracts small molecules into the water. The device brings the water sample into a mass spectrometer instrument that can detect molecular fingerprints.
“When designing the MasSpec Pen, we made sure the tissue remains intact by coming into contact only with water and the plastic tip of the MasSpec Pen during the procedure,” Jialing Zhanf, a research associate, said. “The result is a biocompatible and automated medical device that we are so excited to translate to the clinic very soon.”
The researchers hope to test this technology during oncologic surgeries in 2018. The research was funded by UT Austin through startup funds, the National Cancer Institute of the National Institutes of Health and the Cancer Prevention Research Institute of Texas. It was published in the journal Science Translational Medicine.