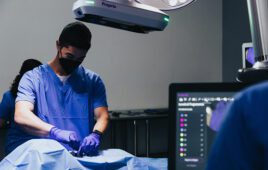
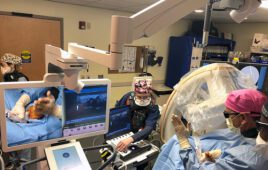
Pixium Vision said today that its Prima miniature wireless photovoltaic sub-retinal implant has been activated in the first five patients.
Pixium Vision said today that its Prima miniature wireless photovoltaic sub-retinal implant has been activated in the first five patients.
The patients all have severe vision loss from atrophic dry age-related macular degeneration. They are enrolled in a three-year clinical study in France to evaluate the safety and function of the Prima chip in eliciting visual light perception, the company said in a statement. All five reported seeing useful light patterns in areas where they had had no remaining light perception. The current study patients will be taught how to interpret their new artificial central macular perception.
The Prima implant is a micro-photovoltaic chip that measures 2-by-2 millimeters wide by 30 microns thick and is equipped with 378 electrodes. Implanted under the retina via a minimally invasive surgical procedure, it acts like a tiny solar panel powered by pulsed, near-infrared light through a miniaturized projector. The projector and a mini-camera are integrated into a pair of augmented-reality-like glasses worn by the patient.
Prima is designed to compensate for severe vision loss from retinal dystrophies such as atrophic dry age-related macular degeneration, the leading cause of severe vision loss and legal blindness in people over the age of 65 in North America and Europe. The condition affects an estimated 12 to 15 million people worldwide, a figure expected to grow given the aging population. The company also plans to test Prima on patients with retinitis pigmentosa.
The Prima feasibility study will be presented during the upcoming international congresses of ophthalmology, Euretina in September and Ever in October in Europe, and the annual congress of the American Academy of Ophthalmology in October in Chicago.
Pixium expects the full set of six-month interim results of the French feasibility study by the end of 2018. This will enable the design of the protocol for the larger multi-centric European pivotal study, which is required for the CE-mark, to commence in 2019. In the United States, the first Prima implantations are expected to be completed in the coming weeks at the University of Pittsburgh (Pa.) Medical Center, the single study center for the U.S. feasibility study.
“The successful consecutive activations of Prima in all five implanted study patients, combined with the preliminary clinical results and progress with the first implanted patients, are very encouraging for the clinical development of Prima,” said Pixium Vision CEO Khalid Ishaque in the statement. “We look forward to reporting on the continued progress of the feasibility study.”