Predicting it will be a “significant market accelerator for proton therapy in the United States,” the Belgian company Ion Beam Applications (IBA) announced FDA clearance its new superconducting accelerator that is an essential component of the Proteus One system for the treatment of cancer.
IBA bills the Proteus One as “smaller, more affordable, easier to install and operate, and…ultimately easier to finance than most common solutions.” The manufacturer indicates that the system is distinguishable from other similar solutions because it is compact and scalable. They note it is a single-room device, as opposed to a more conventional system that necessarily sprawls to consume extensive real estate in a facility.
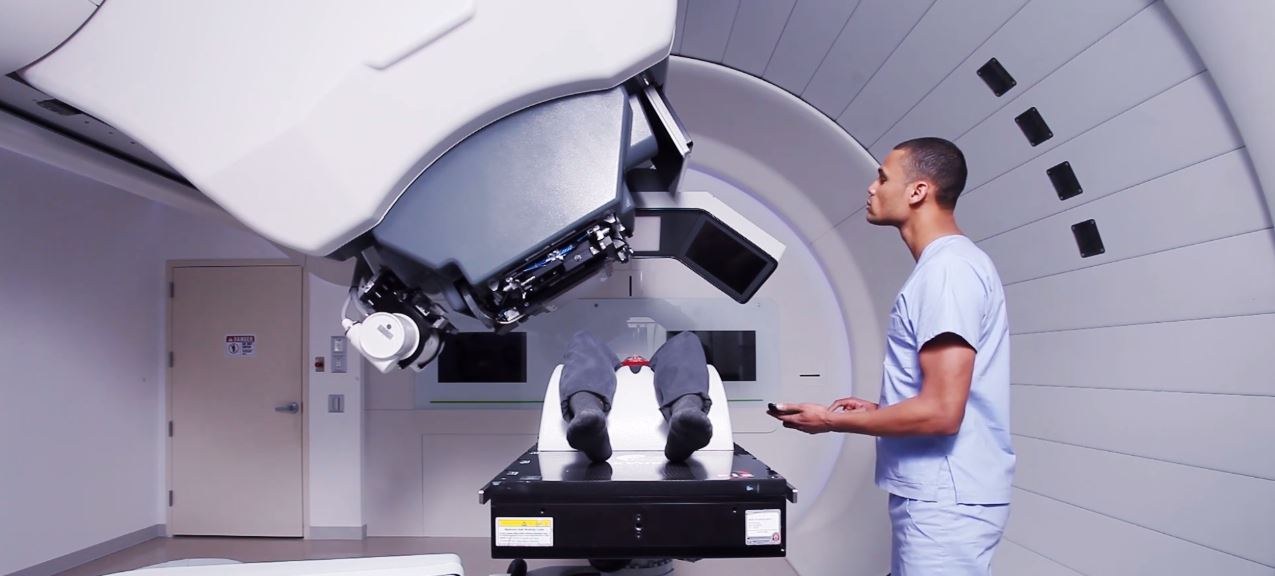
(Image credit: IBA promotional video)
The Proteus One is equipped with Pencil Beam Scanning, described by the company as “ a technology that offers high sub-millimeter precision treatment, which allows delivery of very high levels of conformity and dose uniformity, even in complex-shaped tumors, whilst at the same time sparing the surrounding healthy tissue.”
The company notes it currently has a total of 13 commitments worldwide for delivery of the Proteus One. The superconducting accelerator secured CE mark authorization earlier this year.




