Promise I
 LimFlow’s Promise I trial marked the first human use in the U.S. of a purpose-built percutaneous deep vein arterialization system for treating no-option chronic limb-threatening ischemia (CLTI) patients.
LimFlow’s Promise I trial marked the first human use in the U.S. of a purpose-built percutaneous deep vein arterialization system for treating no-option chronic limb-threatening ischemia (CLTI) patients.
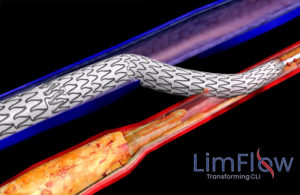
LimFlow’s procedure permanently bypasses non-reconstructible arteries and uses healthier veins as a conduit to create new routes to perfuse tissue in the foot, with the purpose of its Promise I trial to establish safety, effectiveness and feasibility for use in the treatment of CLTI.
The trial enrolled 32 no-option CLTI patients across seven U.S. centers, observing a primary safety endpoint of above-ankle amputation-free survival (AFS) at 30 days, with a secondary endpoint of AFS at six months. Additional secondary endpoints included technical success, vessel patency and wound healing.
LimFlow’s procedure achieved technical success of 97%, with just one failure, while the primary safety endpoint of AFS at 30 days came in at 91% and the secondary endpoint of AFS at 12 months occurred at a rate of 70%. Approximately 75% of wounds healed or were healing.