Researchers at the U.S. Department of Energy’s Brookhaven National Laboratory have created a new imaging technique that allows scientists to probe the internal makeup of a battery during charging and discharging using different X-ray energies while rotating the battery cell. The technique produces a three-dimensional chemical map and lets the scientists track chemical reactions in the battery over time in working conditions.
Getting an accurate image of the activity inside a battery as it charges and discharges is a difficult task. Often even X-ray images don’t provide researchers with enough information about the internal chemical changes in a battery material because two-dimensional images can’t separate out one layer from the next.
“It’s very challenging to carry out in-depth study of in situ energy materials, which requires accurately tracking chemical phase evolution in 3-D and correlating it to electrochemical performance,” said Jun Wang, a physicist at the National Synchrotron Light Source II who led the research.
Using a working lithium-ion battery, Wang and her team tracked the phase evolution of the lithium iron phosphate within the electrode as the battery charged. They combined tomography (a kind of X-ray imaging technique that displays the 3-D structure of an object) with X-ray Absorption Near Edge Structure (XANES) spectroscopy (which is sensitive to chemical and local electronic changes). The result was a “five dimensional” image of the battery operating: a full three-dimensional image over time and at different X-ray energies.
To make this chemical map in 3-D, they scanned the battery cell at a range of energies that included the “x-ray absorption edge” of the element of interest inside the electrode, rotating the sample a full 180 degrees at each X-ray energy, and repeating this procedure at different stages as the battery was charging. With this method, each three-dimensional pixel-called a voxel-produces a spectrum that is like a chemical-specific “fingerprint” that identifies the chemical and its oxidation state in the position represented by that voxel. Fitting together the fingerprints for all voxels generates a chemical map in 3-D.
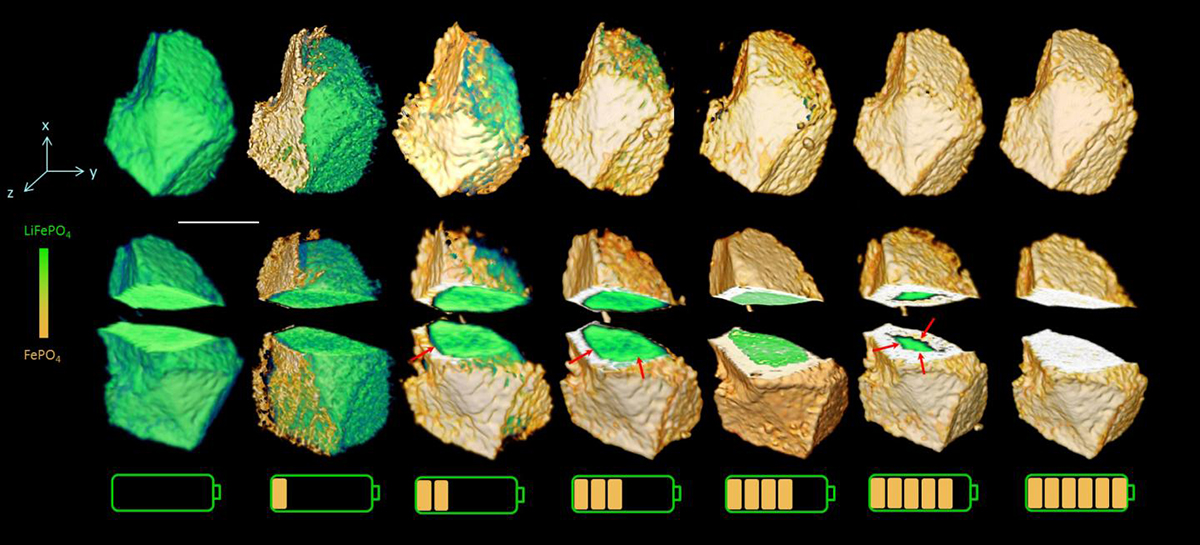
The chemical phase within the battery evolves as the charging time increases. The cut-away views reveal a change from anisotropic to isotropic phase boundary motion. (Credit: Jun Wang)
The scientists found that, during charging, the lithium iron phosphate transforms into iron phosphate, but not at the same rate throughout the battery. When the battery is in the early stage of charging, this chemical evolution occurs in only certain directions. But as the battery becomes more highly charged, the evolution proceeds in all directions over the entire material.
“Were these images to have been taken with a standard two-dimensional method, we wouldn’t have been able to see these changes,” Wang said. “Our unprecedented ability to directly observe how the phase transformation happens in 3-D reveals accurately if there is a new or intermediate phase during the phase transformation process. This method gives us precise insight into what is happening inside the battery electrode and clarifies previous ambiguities about the mechanism of phase transformation.”
Wang said modeling will help the team explore the way the spread of the phase change occurs and how the strain on the materials affects this process.
This work was completed at the now-closed National Synchrotron Light Source (NSLS), which housed a transmission X-ray microscope (TXM) developed by Wang using DOE funds made available through American Recovery and Reinvestment Act of 2009. This TXM instrument will be relocated to Brookhaven’s new light source, NSLS-II, which produces X-rays 10,000 times brighter than its predecessor. Both NSLS and NSLS-II are DOE Office of Science User Facilities.
“At NSLS-II, this work can be done incredibly efficiently,” she said. “The stability of the beam lends itself to good tomography, and the flux is so high that we can take images more quickly and catch even faster reactions.”




