
CorInnova_8M1A8449.jpg William Altman, CEO of CorInnova, presents their EpicHeart device at the 5th Annual Pediatric Device Innovation Symposium hosted by the Sheikh Zayed Institute for Pediatric Surgical Innovation at Children’s National Health System, winning $50,000 in grant money. (Credit: BayAreaEventPhotography.com)
This article is the second of a three-part-series about the recipients of a United States Food and Drug Administration (FDA)-sponsored competition.
EpicHeart, a cardiac assist device for the treatment of end-stage heart failure by CorInnova, was awarded $50,000 at the fifth Annual Pediatric Device Innovation Symposium.
The device is a collapsible, biventricular, soft robotic cardiac device that works by gently squeezing the heart, increasing the input.
Prior to designing the device, the inventor and his team from Texas A&M University researched what materials would be best for allowing the device to fit around the heart, specifically, the inside of the pericardial sac. The final product was a cup-like soft robot made from thin film polyurethane.
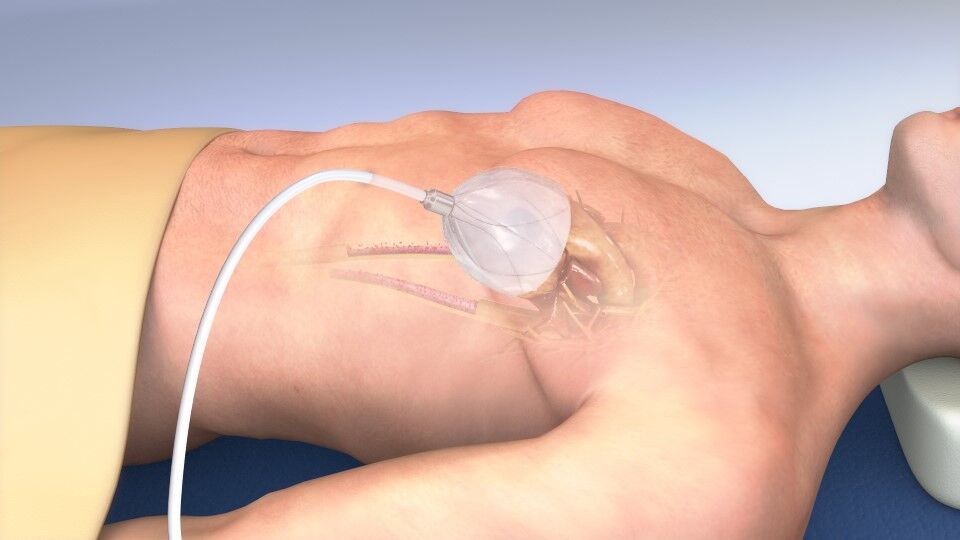
Image of CorInnova’s EpicHeart device (Credit: CorInnova)
The team took caution when designing the nitional wire frame so that it could deploy the delivery tube and travel up and around the heart without needing to use a second deployment device.
Unlike other left ventricular devices needing invasive surgery to implant, the device is inserted using a one-inch tube and implanted into the pericardial space around the heart, accessed between two left-side ribs via a mini left thoracotomy.
Inside of the device is a set of inner polyurethane chambers filled with saline upon implantation. It’s used to fill any small gaps around the heart so that the device exerts an even amount of pressure to the heart from the air-filled chambers around the heart’s surface.
“The actuator augments the power of the heart muscle,” says William Altman, president and CEO of CorInnova. “The device provides biventricular assist when the actuator is active and permits selective withdrawal of assist to wean the patient upon recovery.”
The team also designed an external pneumatic driver to power the device and monitor the heart’s performance.

William Altman, CEO, (right) and Keith Svagerko, VP of Business Development, (left) shown with latest prototype of CorInnova EpicHeart device at CorInnova’s office at JLABS @ TMC in the Texas Medical Center (Credit: Corinnova)
One of the team’s main difficulties during the design process was constructing a soft robotic device capable of compressing the heart, while maintaining the correct curvature of the heart’s surface. They were also worried whether or not the device would function with the heart’s natural rhythm.
“[It] had never been done. It took many years to refine the design to achieve that,” says Altman.
From this experience, the team learned how difficult it is to make a simple design work well.
“[It] is a time-consuming process,” he continues.
If the team is given a chance to design it again, they say they would not change anything. However, they are working on enhancements to help the fail-safe nature of the device.
Altman says that the device was inspired by the fact that surgeons have had to use their hands to squeeze a human heart in distress during surgery. This device replaces the need for medical professionals to pump the heart by hand. The team also thinks the device could be used to heal hearts that have been injured.




