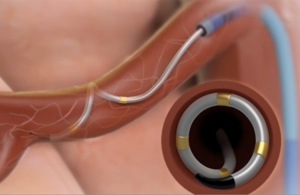
The Symplicity Spyral renal denervation system delivers energy to the nerves leading to the kidneys, which help regulate blood pressure. [Image courtesy of Medtronic]
Medtronic (NYSE:MDT) had to continue its Spyral HTN-ON MED trial into this year after failing to obtain the positive results needed to end enrollment early for the potential hypertension treatment.
During Medtronic’s Q3 earnings call in February, CEO Geoff Martha said the company expects to complete the six-month follow-up to ON MED in the second half of this calendar year. It’s the final piece of information Medtronic needs to submit for FDA approval of its Symplicity Spyral device.
Medtronic’s Symplicity Spyral device delivers energy to the nerves in the wall of the artery leading to the kidney during a minimally invasive outpatient procedure. The idea is to calm overactivity in the nerves that can cause high blood pressure.
Company officials have spoken of a multibillion-dollar market for RDN, estimating global market revenue of more than $500 million by 2026 and $2 billion to $3 billion by 2030.
“After interim data from SPYRAL ON MED trial proved inconclusive, we believe that investors have mostly given up on renal denervation (RDN). But MDT management, for its part, remains bullish on RDN, and the full ON MED data is expected to be revealed in C2H22,” Senior Research Analyst Mike Matson and colleagues said in a report out today.
“We believe that RDN can still be a big opportunity, assuming that the FDA approves the Symplicity Spyral RDN system, which we think might still be possible even with mixed ON MED results.”
The Needham & Co. analysts think the On MED data could be presented at the TCT meeting this September in Boston.




