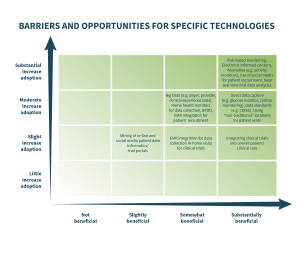
 A rapid increase in the use of wearable technology and an upsurge in social media for patient recruitment are two of the more prevalent clinical trial technology trends, according to a new survey from the Association of Clinical Research Organizations (ACRO).
A rapid increase in the use of wearable technology and an upsurge in social media for patient recruitment are two of the more prevalent clinical trial technology trends, according to a new survey from the Association of Clinical Research Organizations (ACRO).
Full details of the survey may be viewed here.
Other areas where ACRO members identified substantial benefit/increase in adoption were: risk-based monitoring; electronic informed consent (eConsent); and the use of near real-time trial data analytics. Lewis noted that the FDA has already provided guidance on risk-based monitoring and eConsent and the industry has been actively implementing these tools and technologies. ACRO’s members have invested in developing a variety of real-time clinical trial data analytics platforms which are supporting risk-based monitoring, adaptive trial designs and other innovations.
The survey, which formed the basis of ACRO’s comment to the FDA’s public docket, “Using Technologies and Innovative Methods to Conduct FDA-Regulated Clinical Investigations of Investigational Drugs,” was administered by Industry Standard Research.
ACRO
acrohealth.org



