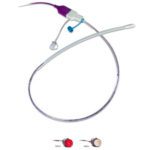
Essure

[Image from Essure]
One study found that the Essure female sterilization device was more than 10 times more likely to require surgery after implantation than those who had laparoscopic sterilization. The British Medical Journal study showed that after one year of having the implant, 2.4% of women who had Essure needed a follow-up surgery, compared to the 0.2% of women who had a minimally invasive tube-tying procedure, according to a British Medical Journal study.
The complaints have been worldwide, with legal complaints being filed in France and Australia in addition to the ones in the U.S. Side effects associated with the device’s failure include depression, dizziness, uterine perforation and autoimmune disorders.
In April 2018, the FDA restricted the U.S. sales of Essure after it concluded that some of the patients were not properly warned of the risks.