Numerous studies in recent years have illustrated the significant difficulty in getting different types of scopes properly cleaned and sanitized following surgery, forcing healthcare facilities to reexamine their processes. With patient safety on the line, it’s especially critical to take exactly the right approach in the sterilization processing department (SPD) and associated areas.
Multiple companies are offering upgraded tools, but PARAMETRIK has a solution that is maybe easiest of all.
“We basically remove the need for surgery centers or hospitals to do their own reprocessing of scopes,” explains Dave Engelke, CEO of PARAMETRIK. “We take over the entire process for them while adding multiple improvements to the standard process.”
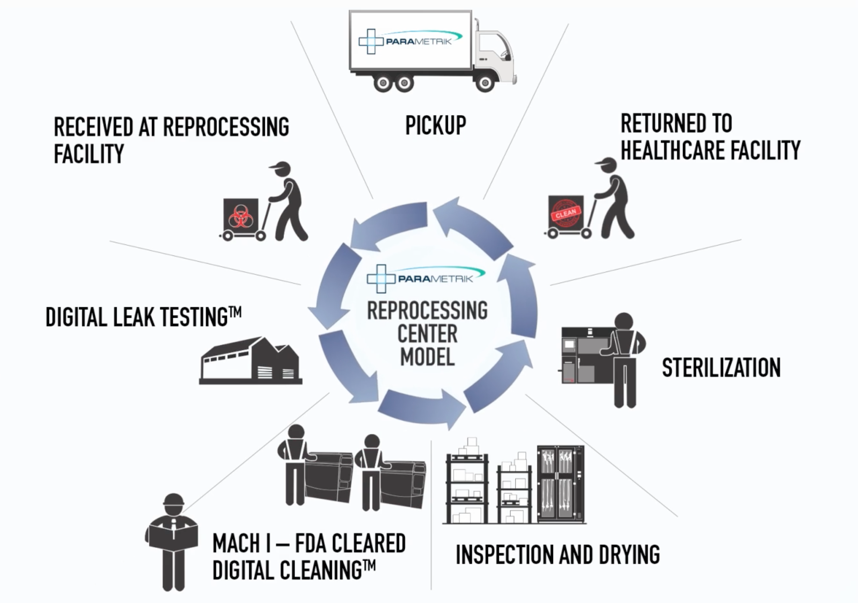
Engelke notes that the protocol for other external sterilization services still relies on a healthcare facility’s SPD unit to do a manual cleaning of the scope, following the manufacturer’s instructions for use (IFU). The PARAMETRIK service begins earlier than that.
“Hospitals are generally relying on their SPD units to do a manual cleaning of the scope following the manufacturer’s IFU, and then they move on to high level disinfection,” says Engelke. “Our service starts immediately post-procedure.”
(Image credit: PARAMETRIK)
According to Engelke, the hospital staff completes a bedside wipe down and channel flush, and then they bag the endoscope in PARAMETRIK’s leak proof biohazard packaging. The packaging includes a proprietary tip protector which promotes retention of moisture on soiled instruments for up to 72 hours. The bagged scope is then placed in an environmentally sealed transport case.
It typically takes no more than a few minutes for endoscopy personnel to complete this post-procedure prep.
“We pick up the box and the rest is up to us,” says Engelke.
Upon arrival at their facility, PARAMETRIK inspects, leak tests, cleans, and terminally sterilizes endoscopes. The devices are returned to the hospital within 24 hours.
PARAMETRIK’s terminal sterile packaging also allows scopes to be stored up to a year. The scopes are ready for the next procedure without any additional prep.
PARAMETRIK uses their own “Parametric Release Instrument Processor” cleaning machine, which is 510(k) approved by the FDA for use on all flexible lumened instruments. The system takes the scopes through a digital cleaning process tracked by machine sensors and software which triggers an alarm if there are any issues completing the cleaning process.
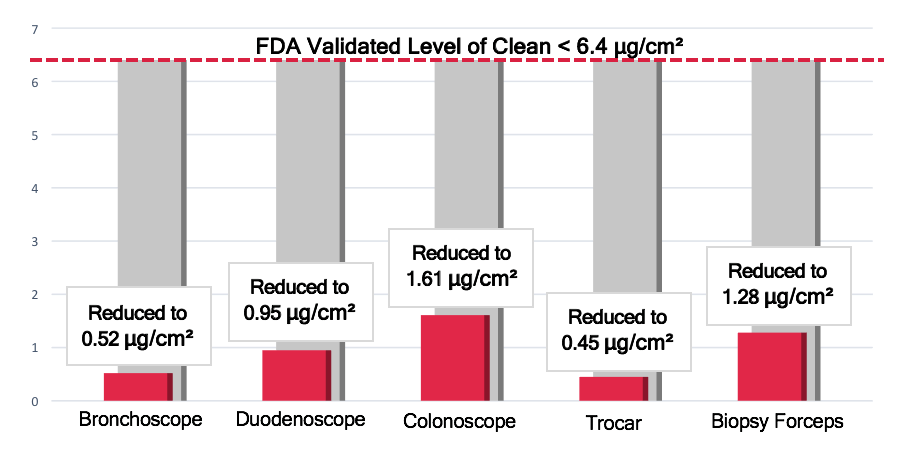
Engleke points to validated studies showing that an endoscope successfully passed through the PARAMETRIK instrument processor will be at least 400 percent cleaner than the current FDA regulation for manually processed scopes.
(Image credit: PARAMETRIK)
“We are cleared by FDA for parametric release,” notes Engelke. “The FDA already rates us as a direct replacement for the manufacturer’s IFU. If a scope is processed through our system and the system diagnostics pass the scope, then it is deemed to be clean to a validated level by the FDA. This is totally unique in the industry. We’re the only one.”
Knowing that healthcare professionals — and their patients — are relying on the scopes covered by PARAMETRIK to be safe, the company is sure to put the devices back in surgeons’ hand in the best possible condition.
“The last piece of our service is a 21st century EO gas sterilization process,” Engleke says. “When we return the scope to the hospital, it’s terminally sterile. It’s not high level disinfected. It’s not just validated clean. It’s terminally sterile.”
By definition, the intensive service-based approach of PARAMETRIK is somewhat limited by geography. The company currently covers New York City and the Research Triangle area in North Carolina. Southern California, Pittsburgh, and the Dallas-Fort Worth regions will be added to the roster soon.




