Heart arrhythmia can be optically addressed (Credit: Johns Hopkins Biomedical Engineering)
Using high-tech human heart models and mouse experiments, scientists at Johns Hopkins and Germany’s University of Bonn have shown that beams of light could replace electric shocks in patients reeling from a deadly heart rhythm disorder.
The findings, published online today in The Journal of Clinical Investigation, could pave the way for a new type of implantable defibrillators.
Current devices deliver pulses of electricity that are extremely painful and can damage heart tissue. Light-based treatment, the researchers say, should provide a safer and gentler remedy for patients at high risk of arrhythmia, an irregular heartbeat that can cause sudden cardiac death within minutes.
This idea springs from advances in the field of optogenetics, in which light-sensitive proteins are embedded in living tissue, enabling the use of light sources to modify electrical activity in cells.
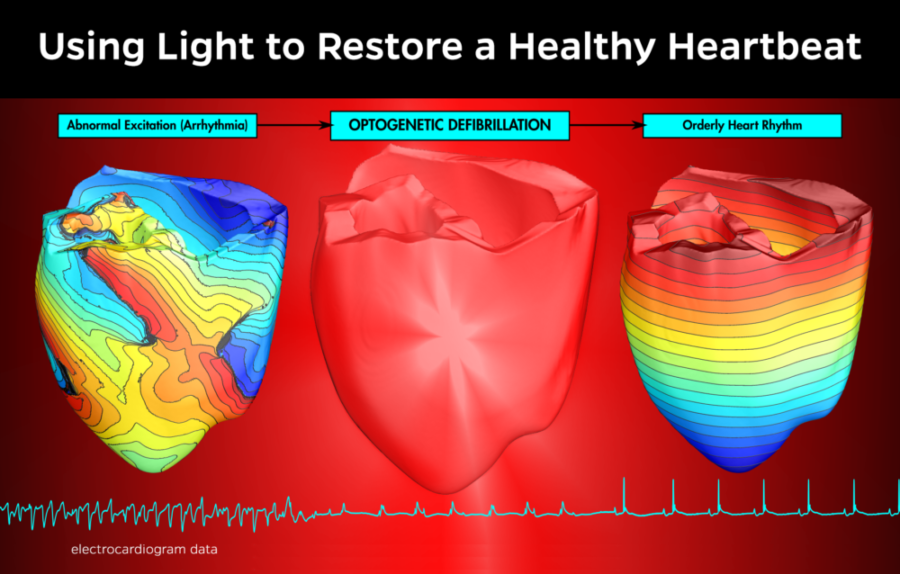
“We are working towards optical defibrillation of the heart, where light will be given to a patient who is experiencing cardiac arrest, and we will be able to restore the normal functioning of the heart in a gentle and painless manner,” said Natalia Trayanova, a professor in JHU’s Department of Biomedical Engineering and a core faculty member in the university’s Institute for Computational Medicine who supervised the research at Johns Hopkins.
To move the new heart treatment closer to reality, the scientists at the University of Bonn and Johns Hopkins focused on two different types of research.
The Bonn team conducted tests on beating mouse hearts whose cells had been genetically engineered to express proteins that react to light and alter electrical activity within the organ. When the Bonn researchers triggered ventricular fibrillation in the mouse heart, a light pulse of one second applied to the heart was enough to restore normal rhythm.
“This is a very important result,” said Tobias Bruegmann, one of the lead authors of the journal article. “It shows for the first time experimentally that light can be used for defibrillation of cardiac arrhythmia.”
To find out if this technique could help human patients, Trayanova’s team at Johns Hopkins performed an analogous experiment within a detailed computer model of a human heart, one derived from MRI scans taken of a patient who had experienced a heart attack and was now at risk of arrhythmia.
“Our simulations show that a light pulse to the heart could stop the cardiac arrhythmia in this patient,” said Patrick M. Boyle, a Johns Hopkins biomedical engineering research professor who was also a lead author of the journal article.
To do so, however, the method from the University of Bonn had to be tweaked for the human heart by using red light to stimulate the heart cells, instead of the blue light used in mice. Boyle, who is a member of Trayanova’s lab team, explained that the blue light used in the much smaller mouse hearts was not powerful enough to fully penetrate human heart tissue. The red light, which has a longer wavelength, was more effective in the virtual human tests.
“In addition to demonstrating the feasibility of optogenetic defibrillation in a virtual heart of a patient, the simulations revealed the precise ways in which light alters the collective electrical behavior of the cells in the heart to achieve the desired arrhythmia termination,” Trayanova said.
Boyle added that this aspect of the study highlighted the important role that computational modeling can play in guiding and accelerating the development of therapeutic applications for cardiac optogenetics, a technology that is still in its infancy.
Junior Professor Philipp Sasse, who is corresponding author of the study and supervised the project at the University of Bonn, agreed that the promising light treatment will require much more time and research before it can become a commonplace medical procedure.
“The new method is still in the stage of basic research,” Sasse said. “Until implantable optical defibrillators can be developed for the treatment of patients, it will still take at least five to 10 years.”




