A wearable heart monitor increases the detection of atrial fibrillation—a heartbeat irregularity that raises the risk of stroke and death—in a study led by the La Jolla-based Scripps Translational Science Institute.
Participants at risk for atrial fibrillation who wore the monitor were diagnosed at three times the rate of control participants, who were checked at regular doctors’ appointments.
It remains to be seen if the higher diagnosis rate translates into better outcomes for patients, said Dr. Steven R. Steinhubl of Scripps Translational Science Institute, who led the study. STSI is affiliated with The Scripps Research Institute.
A total of 5,214 individuals were included in the one-year study. Atrial fibrillation was newly diagnosed in 6.3 percent of the monitored participants, one third of the total. In the control patients, just 2.4 percent were newly diagnosed.
The study was published in the Journal of the American Medical Association. It’s one test of whether digital technology can provide a more accurate diagnosis at less cost than traditional visits to a doctor’s office or the hospital.
By monitoring patients at home with wearable devices, doctors may get a more natural read on a patient’s condition. That’s especially true with intermittent conditions such as atrial fibrillation, Steinhubl said.
It’s also easier on patients, especially those who live in rural areas or otherwise have difficulty reaching a clinic, Steinhubl said. Such studies are usually performed at medical clinics, especially those located in big cities.
“This was designed to be very participant-centric and very pragmatic,” Steinhuble said. “A lot of people who are interested in studies can never participate because they don’t happen to live next to a medical center that’s part of the study.”
The heart information is collected in a “real-world” setting, Steinhubl said, which doesn’t interfere with the routines of either patient or doctor.
The patch is an FDA-approved electrocardiogram (ECG) monitor, the wireless iRhythm ZioXT patch.
Participants received the patches by mail, along with instructions on how to use it. After the test period of up to four weeks was completed, participants mailed back the patches. Then the data saved on the devices was analyzed.
An estimated 2.7 million to 6.1 million Americans have atrial fibrillation, according to the Centers for Disease Control and Prevention. It contributes to an estimated 130,000 deaths per year. Along with increasing risk of stroke, it doubles the risk of death.
Incidence of atrial fibrillation increases with age, along with factors such as high blood pressure, obesity, and diabetes.
But diagnosis is difficult; in about a third of patients the disorder doesn’t cause symptoms.
So the study examined whether these undetected cases could be more effectively diagnosed by actively monitoring people at high risk for atrial fibrillation. They could then be treated before serious effects occur.
STSI researchers worked on the study with Healthagen, a unit of insurer Aetna, and Janssen Pharmaceuticals, Inc. Aetna provided data and analytics to make the study design.
Study participants were Aetna health plan members, who Aetna selected among eligible members who volunteered to take part.
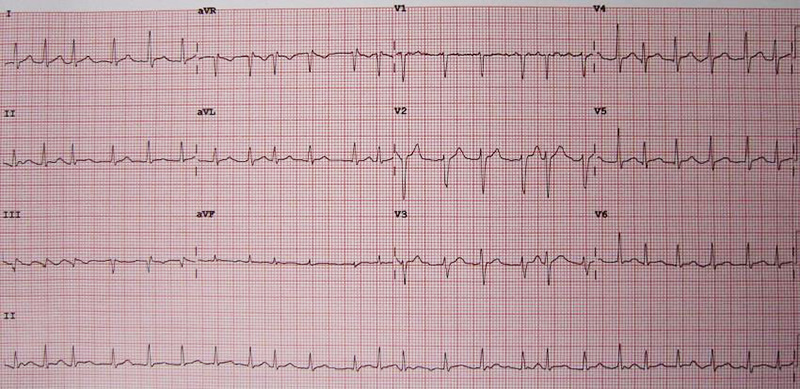
A 12 lead ECG showing atrial fibrillation at approximately 150 beats per minute. Credit: James Heilman, MD/Wikipedia/CC BY-SA 3.0




